Scientists have identified why blood vessels in the brain become overactive in dementia—and how to calm them down. The discovery centers on a single molecule called PIP2, which acts as a natural brake on a protein that controls blood flow. When PIP2 levels drop, that brake fails, vessels become hyperactive, and blood flow suffers. Restore the PIP2, and healthy circulation returns.
The research, published in Proceedings of the National Academy of Sciences by teams at the University of Vermont's Larner College of Medicine, suggests a new angle for treating vascular dementia and related conditions. Rather than targeting the proteins that accumulate in Alzheimer's disease—the approach that's dominated research for decades—this work focuses on restoring the molecular machinery that keeps blood vessels functioning normally.
How the Brain's Blood Flow Gets Disrupted
The brain's blood vessels are lined with cells that contain a protein called Piezo1, which senses physical forces as blood moves through the network. Think of it as a pressure sensor that helps regulate flow. PIP2, a phospholipid found in cell membranes, normally keeps Piezo1 in check. But in dementia, PIP2 levels fall. Without that molecular brake, Piezo1 becomes overactive, disrupting the delicate balance of blood flow and starving brain tissue of oxygen.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxThe connection matters because reduced cerebral blood flow is now recognized as a core feature of vascular dementia, which accounts for 10–20% of dementia cases worldwide. Alzheimer's disease and related dementias currently affect about 50 million people globally, a number climbing steadily as populations age. Most research has focused on protein clumps and inflammation, but this discovery suggests the problem may also lie in the fundamental mechanics of how vessels regulate themselves.
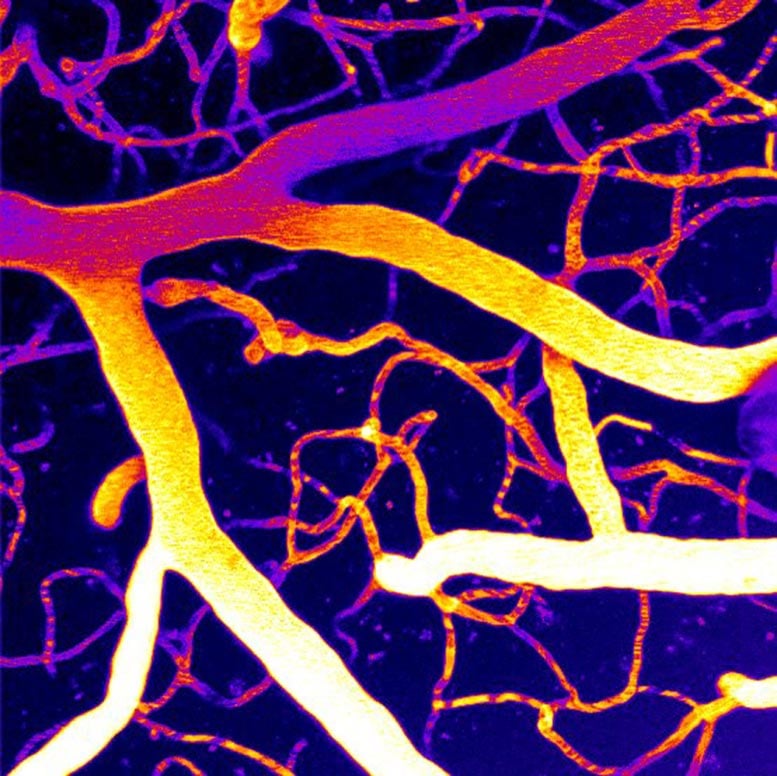
When researchers added PIP2 back into brain tissue in their preclinical models, Piezo1 activity normalized and blood flow improved. "We are uncovering the complex mechanisms of these devastating conditions, and now we can begin to think about how to translate this biology into therapies," says Osama Harraz, the study's principal investigator.
What Comes Next
The immediate question is whether PIP2 restoration could become a drug target. Researchers still need to understand exactly how PIP2 interacts with Piezo1—whether it binds directly to the protein or alters the surrounding cell membrane to limit channel opening. They also need to trace why PIP2 levels drop in the first place during dementia, and whether boosting PIP2 early could prevent the cascade that leads to vascular problems.
If this approach works in human trials, it could offer an alternative pathway for treating dementia that doesn't rely on clearing protein accumulation—something that's proven difficult after decades of effort. The research suggests that sometimes the answer isn't adding something new, but restoring what's already supposed to be there.










