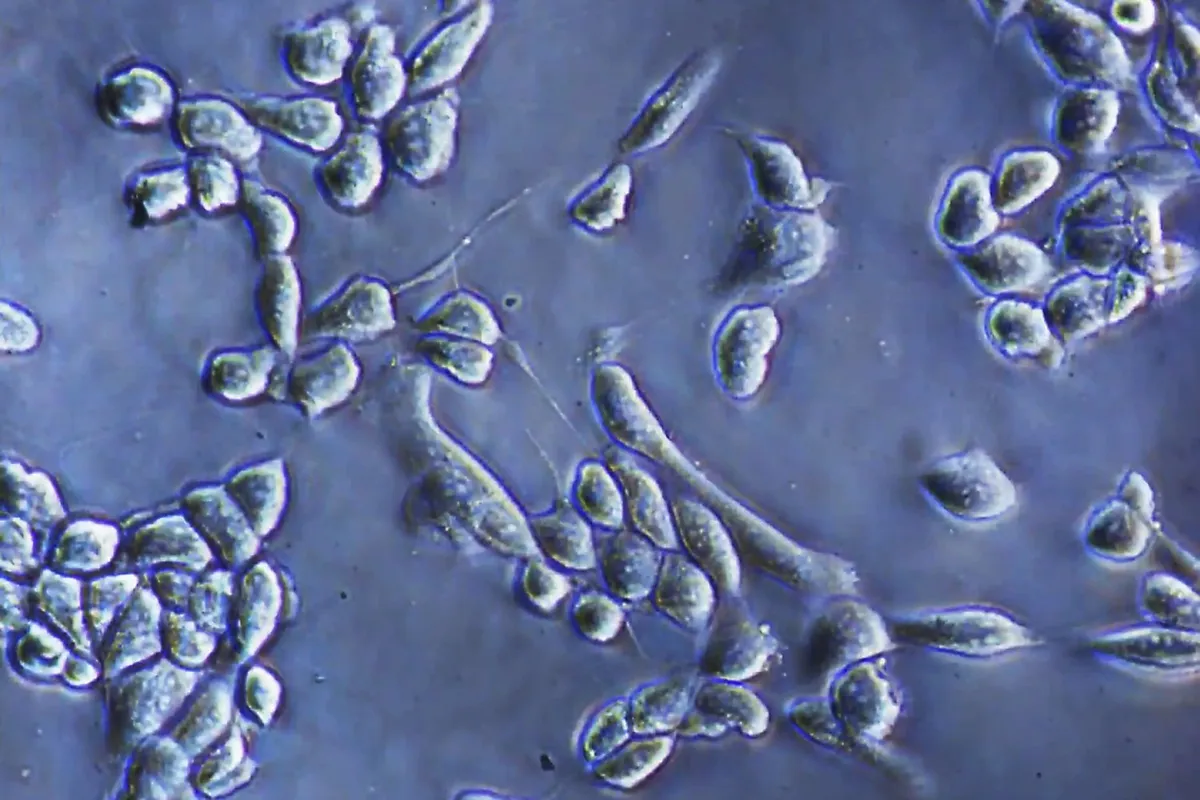
Researchers at the University of Tokyo have built a microscope that does something previous tools couldn't: watch living cells change over hours or days without poisoning them in the process.
The device captures both forward and backward scattered light simultaneously — essentially seeing cells from two angles at once. This expanded view covers an intensity range fourteen times wider than conventional microscopes, letting researchers track both large structures and individual proteins in the same image, at the same time.
"I would like to understand dynamic processes inside living cells using non-invasive methods," says researcher Kohki Horie. The team, including Keiichiro Toda, Takuma Nakamura, and Takuro Ideguchi, spent months solving a deceptively tricky problem: how to cleanly separate two overlapping signals from a single image without introducing noise or cross-contamination. They refined their optics and analysis methods until the two light directions remained distinct.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxWhy this matters comes down to a gap that's existed in cell biology for years. Quantitative phase microscopy can detect large structures — anything above 100 nanometers — using forward-scattered light. Interferometric scattering microscopy, by contrast, can track individual proteins using back-scattered light, but it struggles to see the broader cell context. Most researchers have had to choose one or the other. This microscope bridges that divide.
Watching Cells Change Without Dyes
The absence of fluorescent dyes is the quiet revolution here. Dyes are standard in cell imaging because they glow, making structures easy to spot. But they also interfere with the cell's natural behavior — they can change how cells respond to drugs, how they move, or when they die. A label-free approach means researchers can watch the same cell for days, weeks, or longer, seeing how it genuinely responds to treatment without the dye's interference.
The microscope simultaneously detected movement of large cell structures and tiny particles, while also estimating particle size and refractive index by comparing the two light signals. That last detail — refractive index — hints at a particle's composition or condition, giving researchers clues about what they're actually looking at without needing additional tests.
Keiichiro Toda sees the next frontier clearly. "We plan to study even smaller particles," he says, including exosomes and viruses in various samples. The team also wants to map how cells move toward death, controlling cell conditions and cross-checking findings with other techniques to verify what they're seeing.
For pharmaceutical companies and biotech labs, the implications are concrete: one tool instead of several, shorter analysis pipelines, and the ability to catch subtle structural changes that signal how cells are responding to a drug. The study, published in Nature Communications, suggests this approach could become standard for long-term cell monitoring and drug development testing.
The next step is scaling down — watching even tinier biological structures and seeing whether this method holds up in real-world drug screening and quality control workflows.