For centuries, getting treated for sleeping sickness was almost as bad as, well, sleeping sickness. The go-to IV drug caused a delightful burning sensation and, for a grim 1 in 20 patients, death. Later, an oral treatment required 10 days of pills and brought along a charming host of side effects like vomiting and heart problems. Unsurprisingly, many people just opted out of treatment altogether. Because apparently, that's where we were.
Fast forward to March 2026, and Europe gave the green light to acoziborole. This isn't just another treatment; it's the first single-dose solution for sleeping sickness. We're talking three pills, taken once, and then you're done. Clinical trials found the only side effect was a mild to moderate headache. Which, considering the alternatives, sounds like a spa day.
Dr. Gerardo Priotto, who leads the World Health Organization's (WHO) fight against this particular nightmare, noted that previous treatments were incredibly difficult to administer. Especially, and ironically, in the remote, rural areas where the disease thrives and health services are… limited.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxHow Sleeping Sickness Steals Your Zzz's (and More)
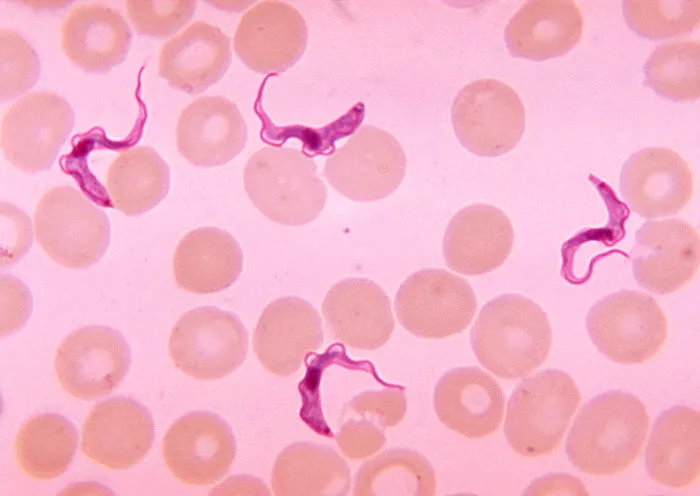
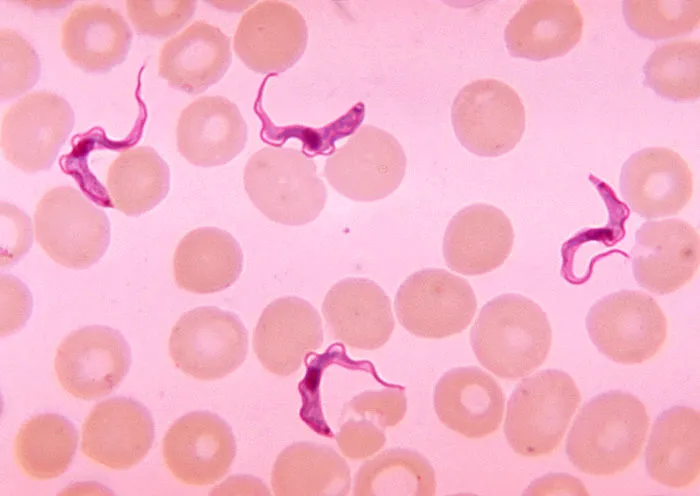
Sleeping sickness spreads via the tsetse fly, a tiny villain that picks up the Trypanosoma brucei gambiense parasite from an infected person and then shares it with the next unfortunate soul it bites. These flies prefer warm savanna woodlands and hanging out near lakes and streams. Which explains why the illness is most prevalent in places where people fish, hunt, and farm — far from the nearest well-stocked pharmacy.
The disease has two stages. First, you get the usual fever and headaches. Then, things get really weird: the parasite invades your brain. Cue confusion, seizures, and a completely flipped sleep schedule — sleepy all day, wide awake all night. Untreated, it’s a one-way ticket to coma and death. Older drugs often only worked on one stage. Acoziborole? It handles both.

At its peak, sleeping sickness infected hundreds of thousands annually. Thanks to tsetse fly control, better testing, and improved access to treatment, that number is now down to about 1,000 cases each year. Nearly two-thirds of those are in the Democratic Republic of Congo (DRC). Let that satisfying number sink in.
Dr. Peter Hotez from Baylor College of Medicine points out that sleeping sickness has historically been a bit of a boomerang, disappearing and reappearing throughout history. The WHO, however, aims to eliminate it entirely by 2030. And health officials are betting on acoziborole to get them there.
Priotto called this single-dose, well-tolerated treatment a "transformative tool," promising to simplify patient care, improve access, and fast-track elimination efforts.

The Drugs for Neglected Diseases Initiative (DNDi), a non-profit founded in 2003, developed acoziborole. They saw that many illnesses in low-income countries were treatable, but drug companies had no financial incentive to develop cures. So, DNDi partnered with Sanofi, with funding from various sources, including the Gates Foundation, to make it happen.
Building a Trial, Literally from Scratch
Dr. Wilfried Mutombo Kalonji, from DNDi, helped run the clinical trials in South Ubangi, a remote province in the DRC. They didn't just run trials; they built the infrastructure for them. He explained that patients often live in areas without electricity or water, so they had to set up everything from training health workers to providing internet, electricity, and safe transport to testing sites. They overcame these challenges, he says, and couldn't have asked for a better solution.
Kalonji is now running a follow-up trial to see if a simple blood test could allow treatment to start the same day a patient is tested. This would skip the longer, more expensive confirmatory tests that are practically impossible to do in the field. Monica Mugnier, a sleeping sickness researcher at Johns Hopkins, notes that the parasite can be a bit of a trickster, sometimes hiding from tests. But if blood tests prove reliable, same-day testing and treatment could dramatically increase the number of patients treated and potentially stop the parasite in its tracks.
Acoziborole still needs the official nod from the DRC Ministry of Health and the WHO. After that, treatment guidelines can be updated, and other countries can approve the drug. But global health experts are also eyeing the funding situation. Hotez points out that sleeping sickness, like many tropical diseases, disproportionately affects the poor, meaning much of the progress relies on international funding. Cuts to global health programs could, rather ironically, limit the drug's reach, even after it gets all its golden tickets.
Dr. Stéphane Hugonnet, who worked on the trials and leads DNDi's sleeping sickness response, remembers that many patients feared treatment due to past horrors. This fear kept people away even when they knew they were sick. Getting acoziborole to the communities that need it will require the same tenacious commitment that first brought case numbers down. Because sometimes, the biggest hurdle isn't the disease, but the memory of its cure.