A year of consistent aerobic exercise didn't just make people feel fitter — their brains actually looked younger on brain scans. Researchers at the AdventHealth Research Institute found that adults who stuck to a structured workout routine for 12 months had brains that appeared nearly a year biologically younger than those who didn't increase their activity.
This matters because brain age isn't just a number on a scan. Scientists use MRI imaging to compare how old a brain looks versus a person's actual age, and that gap — called brain-PAD — has been linked to sharper thinking, stronger memory, and a lower risk of cognitive decline later in life. A brain that looks older than it should can signal weaker physical performance and higher mortality risk. So a measurable shift in the other direction is worth paying attention to.
The 12-month test
The study tracked 130 healthy adults aged 26 to 58. Half were assigned to a moderate-to-vigorous aerobic exercise program: two supervised 60-minute sessions per week in a lab, plus home workouts to reach about 150 minutes total per week. The other half continued their usual routines. After a year, brain scans showed the exercise group's brains had aged about 0.6 years less, while the control group's brains had aged slightly more. Overall, the difference between the two groups approached a full year in favor of exercise.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News Detox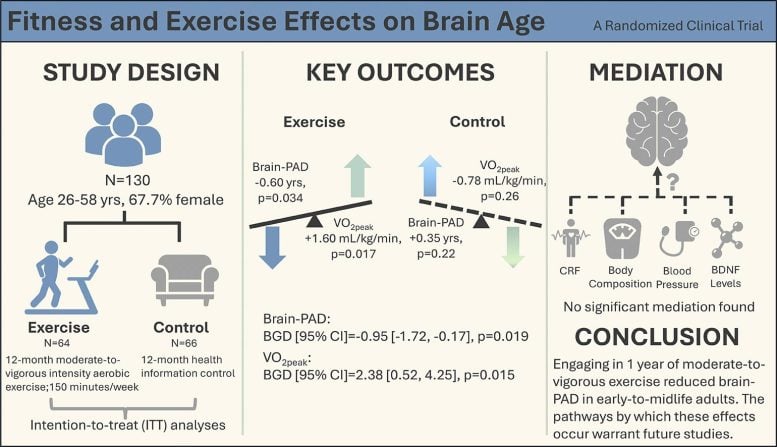
Dr. Lu Wan, the study's lead author, framed it plainly: "A simple, guideline-based exercise program can make the brain look measurably younger over just 12 months." The changes were modest, but Wan noted that even a one-year shift in brain age could compound into meaningful differences over decades.
The mystery of how it works
Here's where it gets interesting: the researchers expected improvements in fitness, blood pressure, or body composition to explain the brain-age shift. They didn't. None of those factors accounted for what they observed. "That was a surprise," Wan said. Exercise appears to be acting through mechanisms researchers haven't yet identified — possibly subtle changes in brain structure, inflammation, vascular health, or molecular factors they're still working to understand.
This gap in knowledge actually underscores something important: we don't need to understand every biological detail to act on what works. The pattern is clear enough.
Why midlife matters most
Most brain-health research focuses on older adults, when age-related changes are already advanced. This study deliberately examined people in their 30s, 40s, and 50s — the stage when preventive strategies may have the biggest long-term payoff. "Intervening in the 30s, 40s, and 50s gives us a head start," said Dr. Kirk Erickson, the study's senior author. "If we can slow brain aging before major problems appear, we may be able to delay or reduce the risk of later-life cognitive decline and dementia."
The researchers are careful to note the study's limits. Participants were healthy and generally well educated, and the sample was relatively small. Larger, longer studies are needed to confirm whether lowering brain-PAD through exercise actually reduces the risk of stroke, dementia, or other age-related diseases. But the signal is there: following current exercise guidelines — 150 minutes per week of moderate-to-vigorous aerobic activity — appears to help keep the brain biologically younger, even in midlife.
The finding offers something increasingly rare in health science: a straightforward answer to a question people actually ask themselves. "Is there anything I can do now to protect my brain later?" Yes. Move regularly, and the evidence suggests your brain will thank you.








