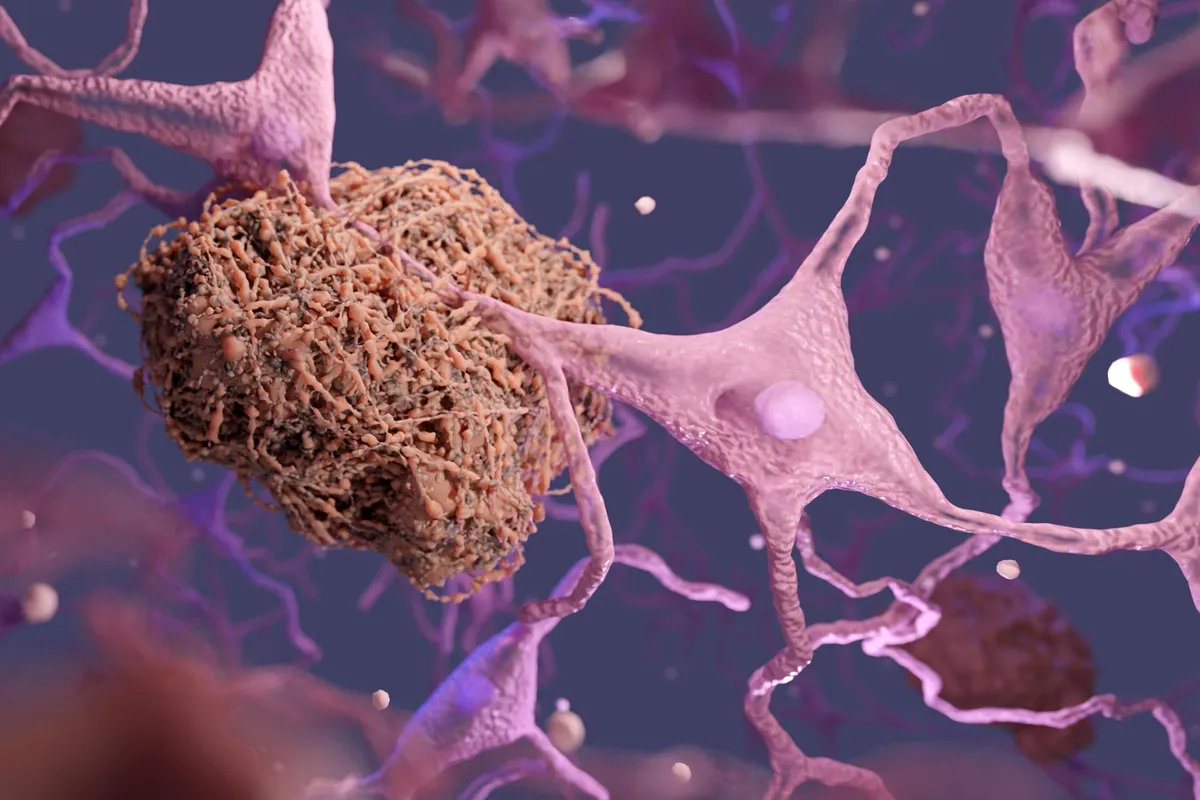
For the first time, scientists have watched the exact moment metal ions trigger the protein tangles that damage Alzheimer's brains—and they've seen how to undo it.
The breakthrough came from Oregon State University chemist Marilyn Rampersad Mackiewicz and her team, who used a technique called fluorescence anisotropy to observe the chemical dance between metals and amyloid-beta proteins in real time, second by second. Most Alzheimer's research has only shown the end result—the clumps that choke neural communication. This work reveals the mechanism itself, which changes everything about how researchers might design treatments.
"We developed a method that lets us observe those interactions live, second by second, and directly measure how different molecules interrupt or reverse them," Mackiewicz explained. "It shifts the question from 'does something work?' to 'how does it work, and when?'"
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News Detox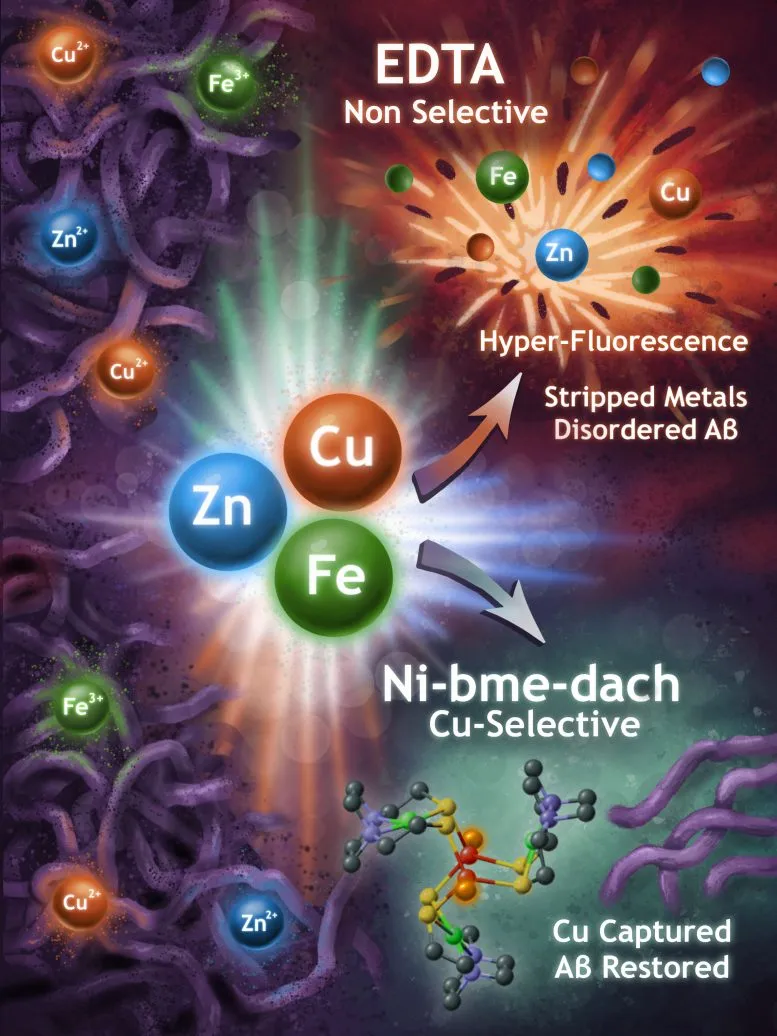
Alzheimer's kills more people over 65 than any other neurodegenerative disease. The clumping of amyloid-beta proteins is the hallmark—these tangles disrupt the electrical chatter between brain cells that makes memory and thought possible. The puzzle has always been why this happens. The answer, it turns out, involves copper and other metal ions. In normal brains, these metals are essential. But when levels become imbalanced, they act like glue, binding amyloid-beta proteins together into the toxic clumps associated with cognitive decline.
The Specificity Problem
The team tested two different chelators—molecules that bind tightly to metals (the name comes from the Greek word for claw). One chelator was broad-spectrum: it grabbed metals indiscriminately, binding both the harmful copper ions and the ones the brain actually needs. The second was a sniper. It showed a strong preference for copper specifically, the metal most implicated in amyloid aggregation.
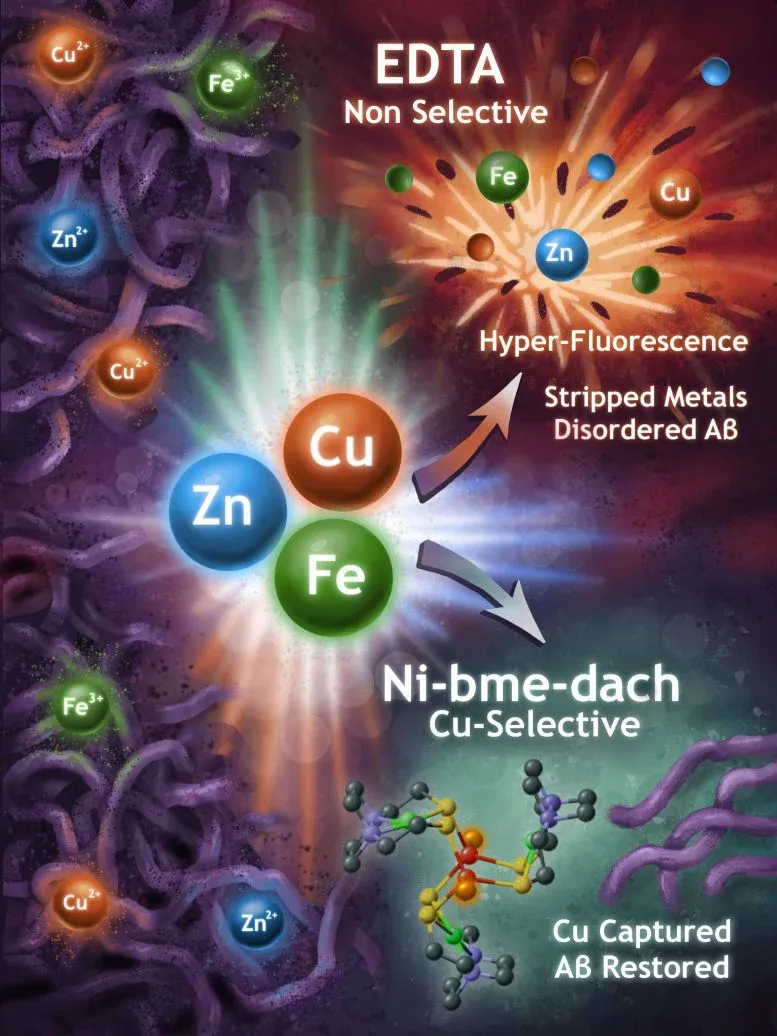
This distinction matters enormously. Many Alzheimer's treatments have failed because they target too broadly, disrupting the brain's normal metal chemistry in the process. By identifying which metals actually drive the problem, researchers can now design drugs that are surgical rather than sledgehammer—removing copper without collateral damage to the rest of the brain's mineral balance.
The original analysis here reveals something the source article only hints at: this work explains why previous drug candidates have flopped. It's not that the science was wrong; it's that researchers didn't understand the mechanism well enough to hit the right target. Now they can.
Mackiewicz's team plans to test their findings in cellular and preclinical models next—moving from the petri dish toward something that might eventually reach patients. Clinical treatments are likely years away. But for the millions of families watching a loved one fade into Alzheimer's, this kind of mechanistic clarity is the foundation that breakthroughs are built on. Understanding the "how" is the first step toward stopping it.