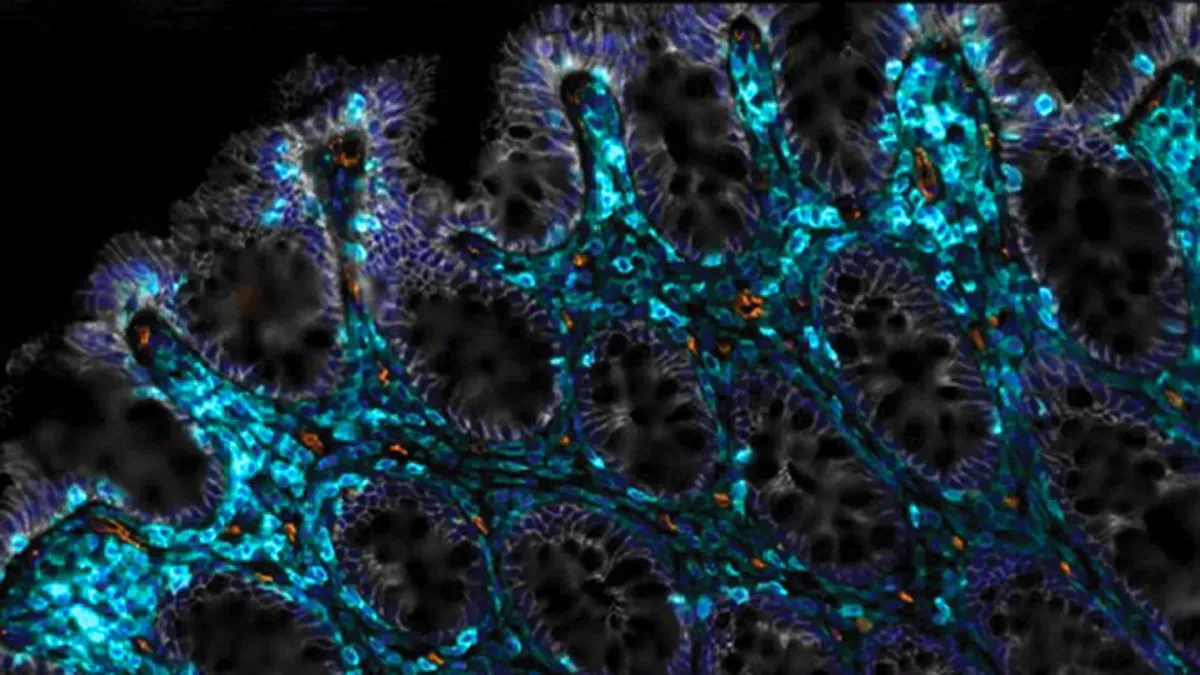
Your gut lining replaces itself every few days under normal conditions. But radiation, aging, or certain medications can stall that process entirely, leaving you with chronic inflammation and food sensitivities that seem to appear out of nowhere.
Researchers at Cold Spring Harbor Laboratory have found something unexpected: a cancer immunotherapy called CAR T-cell therapy can restart intestinal repair at the cellular level. In mice exposed to radiation damage, a single dose of these modified immune cells restored gut function for at least a year—far better than mice that received no treatment.
How This Works
The intestinal lining is deceptively simple: a single layer of cells that acts as both a gateway and a gatekeeper. It lets nutrients through while blocking harmful bacteria and toxins. When this barrier breaks down—what some call leaky gut—inflammation rises and food tolerances narrow. Cancer patients receiving pelvic or abdominal radiation face this problem regularly. So do many aging adults, though the connection isn't always obvious.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxThe CSHL team tested what they call anti-uPAR CAR T cells. These are immune cells engineered to target a specific protein involved in tissue repair. When introduced to radiation-damaged intestines in mice, they didn't just patch the problem—they seemed to reactivate the gut's own regeneration machinery. The effect persisted long-term, suggesting the treatment wasn't a temporary fix but a genuine restart.
Graduate student Onur Eskiocak and his colleagues also tested the approach on human intestinal cells grown in the lab. The same regenerative effect appeared. The precise biology remains unclear—exactly which cellular signals these CAR T cells are triggering—but the pattern is consistent.
What makes this significant is the source. CAR T-cell therapy is expensive and complex, designed for serious cancers. Repurposing it for age-related gut decline suggests the underlying mechanism might be powerful enough to justify that complexity. More likely, future treatments will use what researchers learn here to develop simpler, cheaper approaches.
Assistant Professor Semir Beyaz frames it carefully: "This is one good step toward a long journey in understanding how we can better heal the elderly." That's the realistic view. This isn't a cure ready for your medicine cabinet. It's a proof of concept—evidence that the aging gut's inability to repair itself might not be inevitable.
The next phase involves human trials, probably starting with cancer patients who've already suffered radiation damage. If those work, the research might eventually point toward treatments for the broader population of aging adults whose digestive complaints often get dismissed as normal decline.