Your gut lining replaces itself every few days under normal conditions. But as you age, or after cancer radiation treatment, that renewal process stalls. Inflammation creeps in. Digestion falters. The lining becomes porous — what doctors call leaky gut — and your body struggles to absorb nutrients properly.
Researchers at Cold Spring Harbor Laboratory have now shown that a form of immune therapy can restart that healing process, with effects lasting at least a year from a single treatment.
The strategy targets senescent cells — aging cells that have stopped dividing but refuse to die. They linger in tissues, releasing inflammatory signals that interfere with normal repair. As we age, these cells accumulate throughout the body and have been linked to diabetes, dementia, and other age-related diseases. In the gut, they're particularly problematic because they block the intestinal epithelium's natural ability to regenerate.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxClearing the Debris
Corina Amor Vegas, an assistant professor at CSHL, has spent years studying senescence. Her lab developed specialized immune cells called anti-uPAR CAR T cells — a type of engineered immune cell originally designed to fight certain cancers. The team wondered: what if these cells could selectively remove senescent cells from the gut and let healing resume?
Working with colleague Semir Beyaz and graduate student Onur Eskiocak, they injected these CAR T cells directly into the intestines of both young and older mice. The results were consistent across ages. "They're able to absorb nutrients better," Amor Vegas reported. "They have much less inflammation. When irritated or injured, their epithelial lining is able to regenerate and heal much faster."
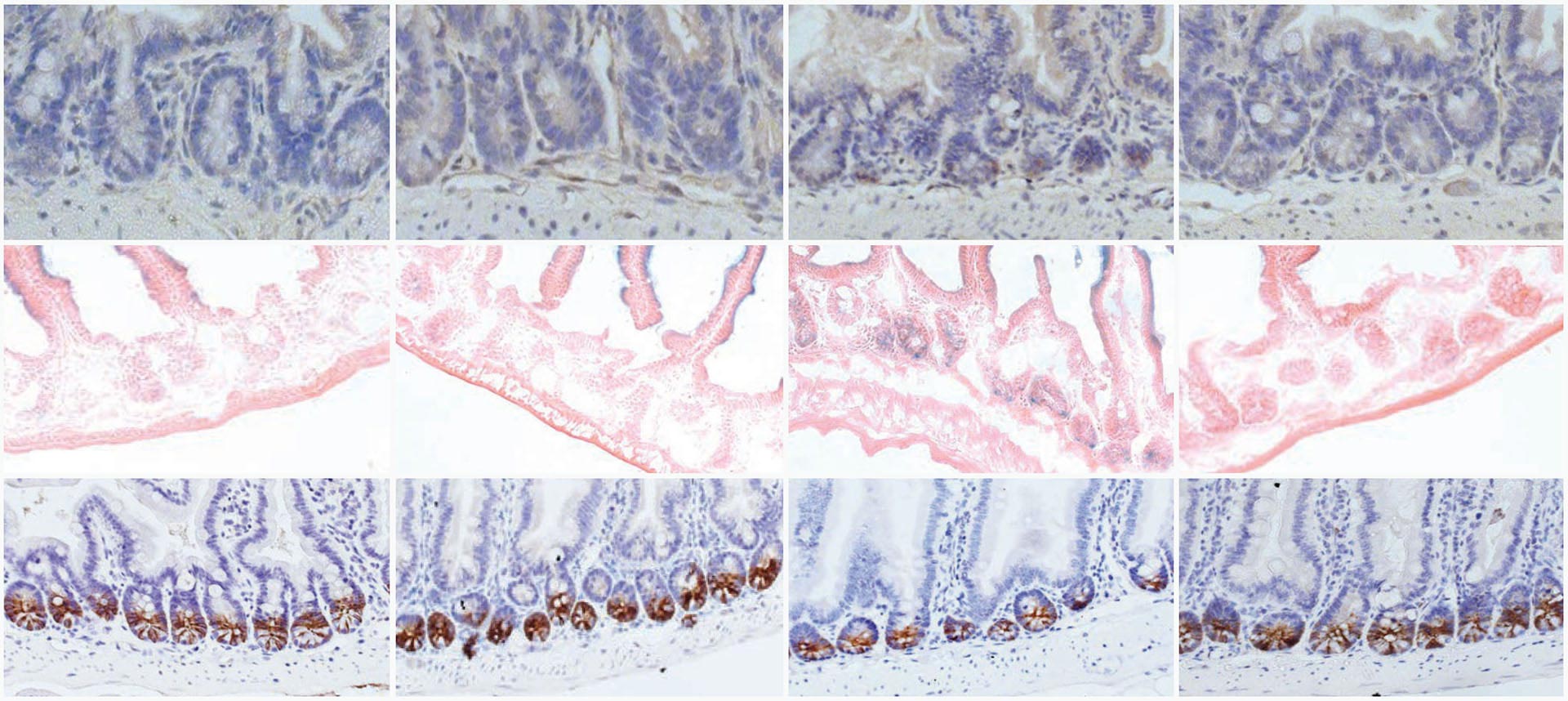
Treating the intestinal linings with uPAR-targeting CAR T cells improved gut health in both young mice (left) and older animals (right). Treated intestines (shown in the second and fourth columns) exhibited reductions in uPAR cells (top row), as well as reductions in damaged senescent cells (middle row) and increases in healthy stem cells (bottom row). Credit: Amor Vegas & Beyaz labs/CSHL
The team then tested whether this approach could repair damage from radiation — a real problem for cancer patients undergoing pelvic or abdominal therapy. They exposed mice to the same radiation dose that damages human intestinal cells, then treated some with CAR T cells. The treated mice recovered far more effectively. More striking: a single dose continued to improve gut health for at least one year.
When the researchers tested the approach on human intestinal and colorectal cells in the lab, they saw the same regenerative signals. The exact mechanisms are still being worked out, but the evidence is encouraging. "This is one good step toward a long journey in understanding how we can better heal the elderly," Beyaz said.
The findings suggest that CAR T-cell therapy — already proven in cancer treatment — might eventually help people recover from age-related gut decline and the intestinal damage that comes with radiation therapy. The next phase would be moving toward human clinical trials.