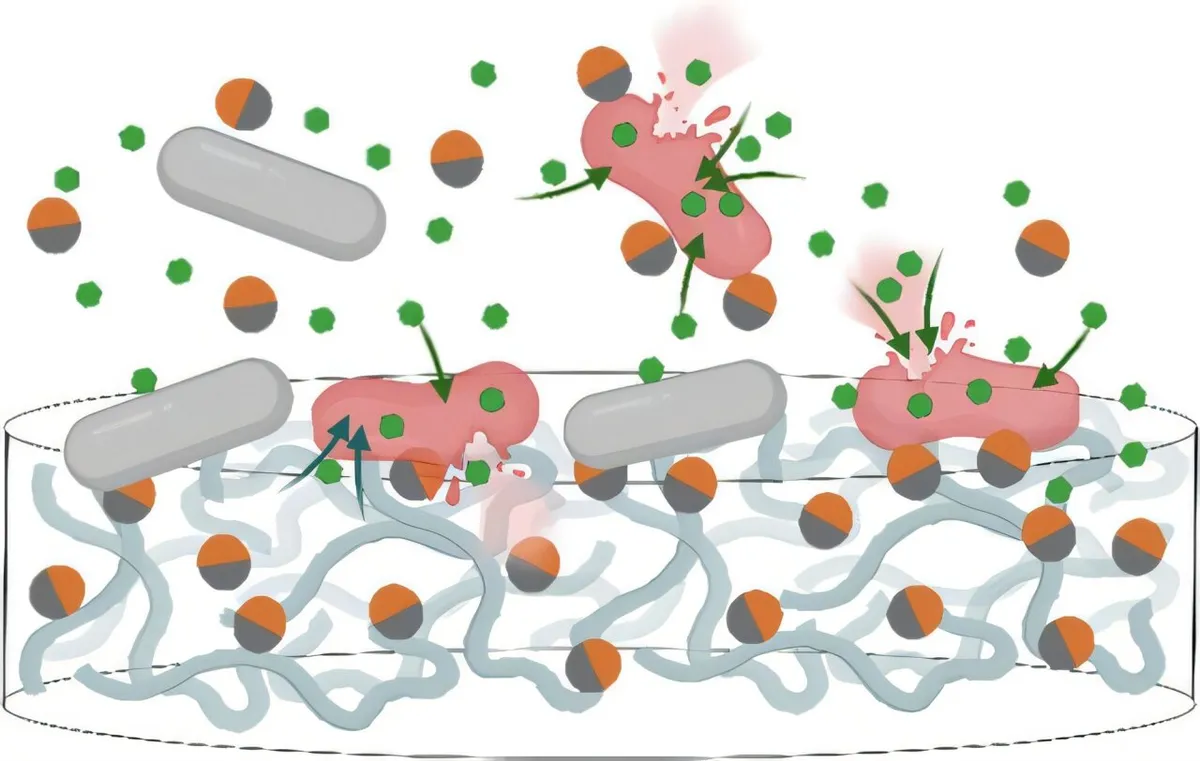
Antibiotic resistance has become one of modern medicine's most stubborn problems. Bacteria evolve defenses faster than we can develop new drugs, and the pipeline for novel antibiotics has slowed to a trickle. But researchers at Washington University have found a different path: instead of searching for entirely new weapons, they're supercharging the old ones.
A team led by chemistry professor Yan Yu created nanoparticles so small they're measured in nanometers—invisible to the naked eye—but with an outsized effect on drug-resistant bacteria. The particles, called Janus nanoparticles after the Roman god with two faces, are engineered with two distinct sides. One side is coated in positively charged molecules that stick to bacterial cell walls. The other side carries water-repelling molecules that rupture those walls. Together, they breach the bacteria's defenses in a way neither component could manage alone.
When the bacterial cell wall breaks open, antibiotics that previously couldn't penetrate suddenly find a way in. In lab tests, this combination killed drug-resistant strains of E. coli and A. baumannii far more efficiently than antibiotics alone. One of the A. baumannii strains was collected directly from a hospital—a real-world sample of the kind of superbug that currently leaves clinicians with few options.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News Detox"The nanoparticles supercharged the antibiotics," Yu said. "This research could point to a new way to give new life to old anti-bacterial drugs."
What makes this approach compelling is that it doesn't require waiting years for entirely new antibiotics to be discovered and approved. Instead, it takes drugs we already have—medicines that have been used safely for decades—and restores their power. The bacteria haven't yet evolved a defense against being physically punctured by nanoparticles, which suggests this strategy might stay ahead of resistance longer than previous approaches.
The next step is moving from lab to clinic. Yu is planning collaborations with colleagues across Washington University, including clinicians at WashU Medicine, to test whether Janus nanoparticles can help in real hospital settings—perhaps starting with wound infections, where topical application would be straightforward. She's also exploring how to manufacture these particles at scale and customize them for different bacterial targets.
The timeline remains uncertain, and these results are early. But the fundamental insight is solid: sometimes the solution to an old problem isn't a new invention—it's a clever way to make what already exists work better.