Sexual health care just got quieter, faster, and more private. The FDA has approved at-home testing for gonorrhea, chlamydia, and trichomoniasis—meaning you can now screen yourself in your bedroom instead of sitting in a clinic waiting room. At the same time, two new oral medications for gonorrhea hit the market, ending a decade-long stretch where doctors had essentially one antibiotic left in their arsenal.
For public health experts, the timing feels almost miraculous. STD rates have been climbing for years, and the pandemic scrambled testing and treatment systems nationwide. Now, for the first time in a while, there's momentum shifting the other direction.
"Sexual health can be stigmatized and people can be hesitant about testing," says Dr. Ina Park, a sexual health specialist at UC San Francisco. "Now we have a lot of options for patients who may be wary of going into a provider's office."
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxThe Testing Breakthrough
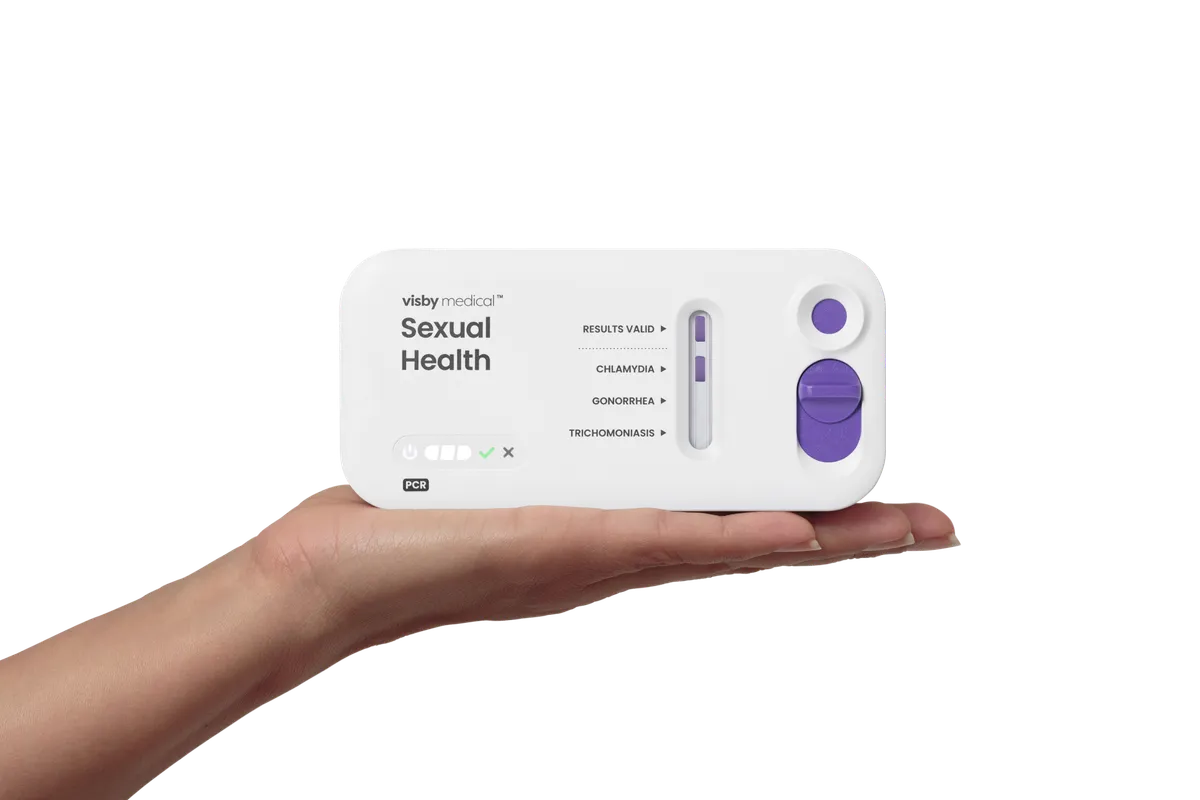
Last year, the FDA approved Visby Medical's at-home test that screens for three infections with a vaginal swab and a small diagnostic device. You collect the sample, photograph the results with an app, and can have a telehealth consultation and prescription within six hours. The accuracy hovers around 98 percent—matching traditional lab tests—and costs $150.
The convenience addresses a real problem: patients with positive results often vanish from the system, never filling prescriptions or following up. A streamlined path from test to treatment closes that gap.
Teal Health's HPV self-collection kit works differently—you collect a sample with the Teal Wand and mail it to a lab—but it marks the first time federal guidelines officially endorse self-collection for cervical cancer screening. That's a milestone for prevention.
What makes these tools powerful isn't just privacy. It's removal. Testing at home means no scheduling around work, no childcare logistics, no sitting in a waiting room wondering who might recognize you. For people already hesitant about sexual health screening, that friction matters.
Treatment Gets Options
On the medication side, the FDA approved two new oral drugs for gonorrhea—Nuzolvenc and Bluejepa—both taken by mouth instead of injected. That matters because gonorrhea has become increasingly resistant to antibiotics. For years, doctors relied on one class of drugs with dwindling effectiveness. "We were down to one class of antibiotics recommended to treat gonorrhea and we had no other good options," Park says. "So to have two new options in the same year is very exciting."
These aren't miracle cures, but they're proof that the infection hasn't outpaced medicine entirely. Yet.
Early Signs of Turning the Tide
CDC provisional data shows gonorrhea cases dropping for three consecutive years. Syphilis and chlamydia are declining too. Public health experts point to multiple factors: less sexual activity among younger populations, growing interest in preventive antibiotics, and wider access to screening. Home testing is still new, but it's already reshaping behavior.
The catch: not everyone can access these innovations equally. A $150 test is convenient for someone with disposable income. For lower-income individuals, it's a barrier. And with recent CDC funding cuts, there's real fear that the people who need these tools most will be the last to reach them.
Park's optimism is genuine but guarded. "I'm feeling very optimistic about the fact that people have more testing options and also that we now have access to new drugs," she says. "What I fear is these cuts to public health are going to decrease access to sexual health care for populations who can least afford to take advantage of these new options."
The question now is whether these innovations stay tools for the privileged or become infrastructure for everyone. The science has moved. The access hasn't caught up yet.