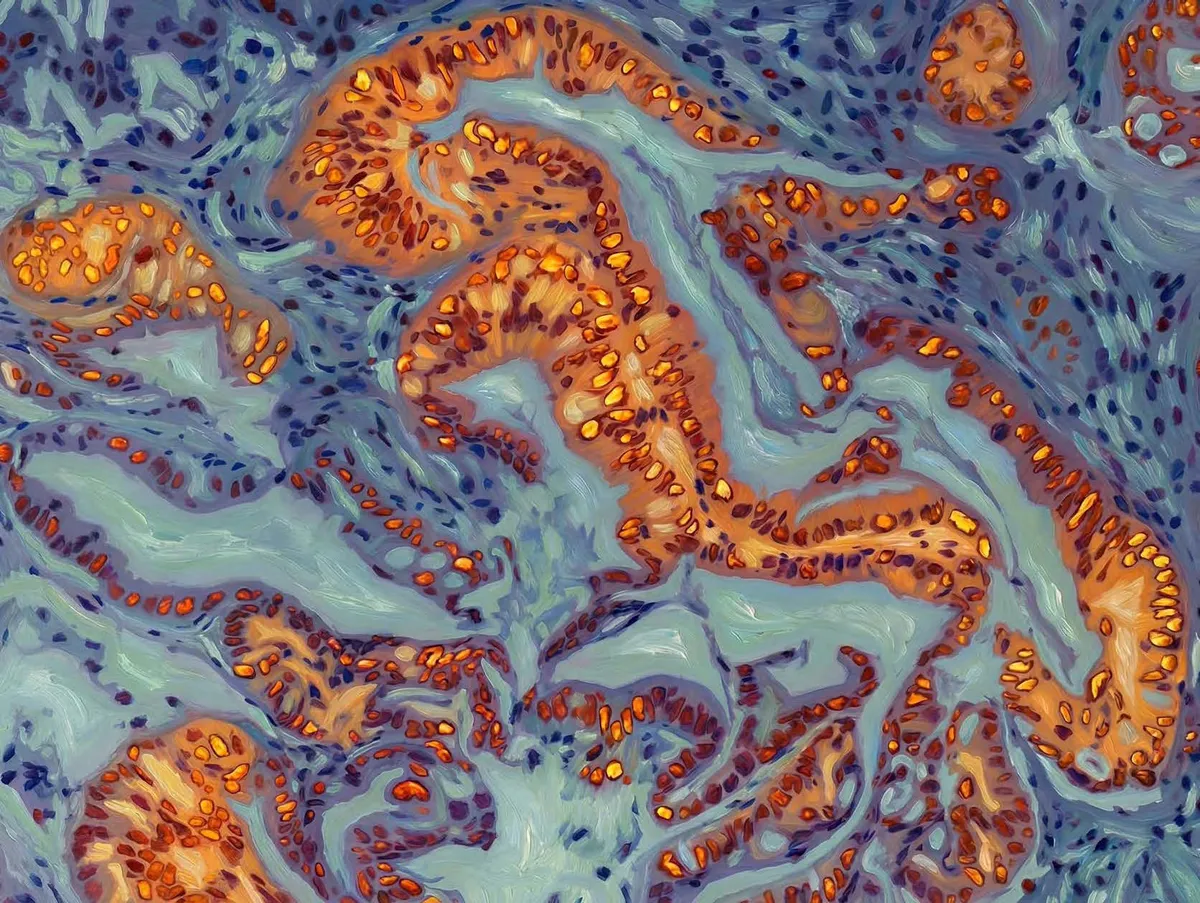
Pancreatic cancer cells have a secret: they can change their identity to escape chemotherapy. Researchers at Duke-NUS Medical School have now found the molecular dial that controls this shape-shifting—and shown how to flip it back.
The discovery, published in the Journal of Clinical Investigation, identifies a gene called GATA6 that acts as a kind of structural blueprint. When GATA6 is active, cancer cells stay organized and respond to drugs. When it gets suppressed, cells become chaotic and resistant. The breakthrough is that scientists can now block the suppression, potentially restoring a tumor's vulnerability to existing treatments.
Why This Matters for a Disease That Kills Fast
Pancreatic cancer is brutal. It's the ninth most common cancer in Singapore but the fourth leading cause of cancer death—meaning it kills faster than its frequency would suggest. Most patients are diagnosed late, when the disease has already spread, and chemotherapy offers only limited help. For decades, doctors have watched tumors simply shrug off the drugs.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxThe reason is cancer cell plasticity. Pancreatic tumors exist in two main states: classical (organized, drug-responsive) and basal (chaotic, drug-resistant). The cells aren't locked into one form. They drift between states like water finding cracks. Once they shift to basal, they're nearly untouchable.
What scientists didn't understand was the mechanism driving that drift. "We have known that pancreatic cancer cells can switch between these two states," explains Professor David Virshup, who led the study. "What we didn't understand was the mechanism driving that switch."
Now they do. The pathway works like this: KRAS, a gene mutated in nearly all pancreatic cancers, constantly tells cells to grow. It sends signals through a protein called ERK. When ERK activity spikes, it protects another protein that suppresses GATA6. GATA6 levels drop. Cells lose structure. Chemotherapy stops working.
The researchers demonstrated that blocking the KRAS-ERK pathway does the reverse. GATA6 bounces back. Cells reorganize. And crucially, when this pathway blocker was combined with standard chemotherapy, the effect was stronger than either drug alone—but only when GATA6 was present. This explains why some patients respond better to treatment than others: it's partly about GATA6 levels.
A Bridge to Existing Treatments
What makes this finding immediately actionable is that it doesn't require inventing new drugs. Multiple KRAS and ERK inhibitors already exist and are in clinical trials. This research provides the biological logic for combining them with chemotherapy—something oncologists can test now.
The insight also reaches beyond pancreatic cancer. Many tumors driven by KRAS mutations—including lung and colorectal cancers—show similar patterns of cell plasticity and treatment resistance. Understanding how cancer cells switch states gives researchers a template for designing combination treatments across multiple cancer types.
Clinical trials testing these combinations are already underway. The next phase is identifying which patients will benefit most—likely those whose tumors still carry detectable GATA6, or those where the KRAS-ERK blocker successfully restores it. That's the work of the next few years.