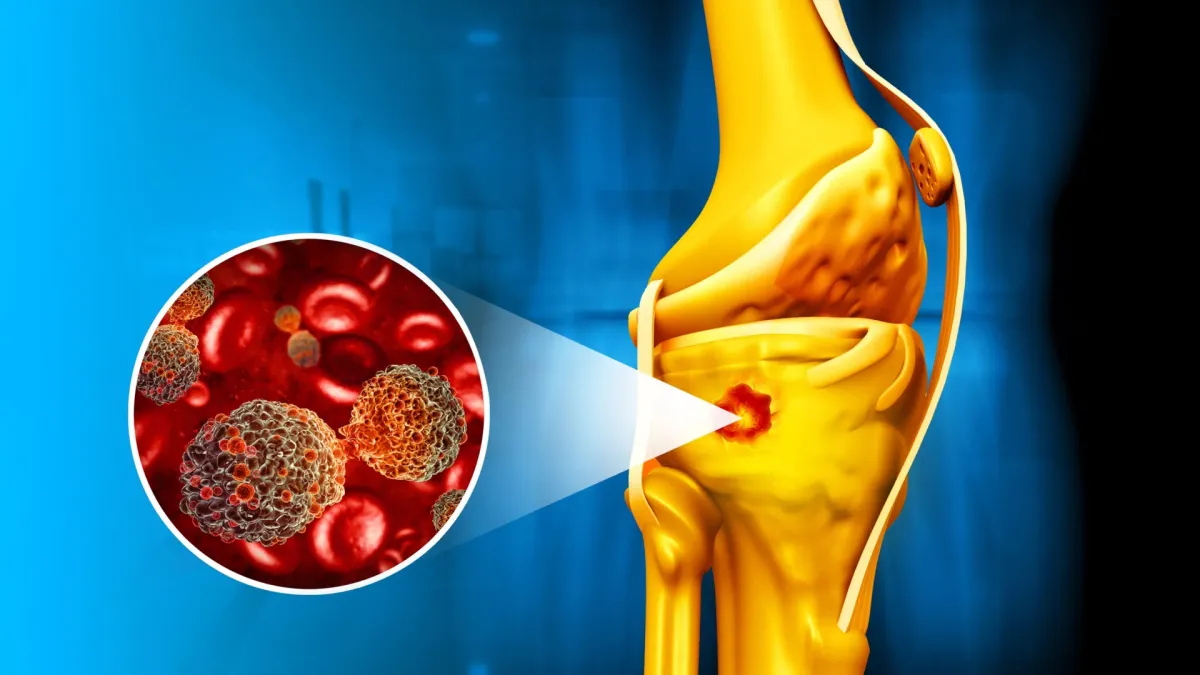
Researchers in Brazil and Portugal have engineered a material small enough to be injected that does something previously impossible: it kills cancer cells in bone while simultaneously triggering the bone to rebuild itself.
The breakthrough works through a deliberately layered design. At the core sits iron oxide — a magnetic material that heats up when exposed to a magnetic field outside the body, essentially cooking cancer cells from within. Wrapped around that core is a thin shell of bioactive glass, which chemically signals to the body's bone-building cells to get to work.
"Magnetic bioactive nanocomposites are very promising for bone cancer therapy because they can simultaneously ablate tumors through magnetic hyperthermia and support new bone growth," said Dr. Ângela Andrade, the lead researcher.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxIn laboratory tests, when the nanocomposite was placed in a solution mimicking body chemistry, it rapidly formed apatite — the mineral that makes up the solid structure of natural bone. The team found that versions with higher calcium content performed best, responding more strongly to magnetic fields and triggering faster mineral formation.
Why This Matters for Patients
Bone cancer is brutal partly because treatment forces an impossible choice: remove the tumor and lose function in that bone, or try to preserve it and risk the cancer spreading. Current approaches typically require surgery to cut out the tumor, followed by separate procedures to rebuild the bone — if that's even possible.
This dual-action approach could collapse that timeline into a single minimally invasive procedure. A surgeon could inject the nanocomposite directly into the tumor site, apply an external magnetic field to heat and destroy cancer cells, and then let the material's glass coating coax the bone to regenerate around it. One treatment. Two outcomes.
The research is still in the lab-and-simulation phase — the team tested it in body-fluid solutions, not yet in living bone. But the results suggest the material bonds well enough with bone tissue that clinical trials could follow within a few years. What makes this particularly promising is that both the iron oxide core and bioactive glass coating are already used separately in medical devices, so the safety profile is partially established.
Dr. Andrade notes that the findings reveal how surface chemistry and material structure influence performance in ways that could guide development of even more sophisticated multifunctional materials. The next step is animal testing, followed by the long path toward human trials — but the concept is now proven.