Colorectal cancer is supposed to be a disease of older age. For decades, it was. But over the past 30 years, something shifted: while traditional colorectal cancer cases have steadily declined, diagnoses in people under 50 have climbed sharply. Today, roughly 12% of all colorectal cancer cases in the United States occur in younger adults, and researchers still weren't sure why.
Now a team at UT Southwestern Medical Center has identified a potential culprit: inflammation doesn't just trigger cancer cells directly—it fundamentally changes the tissue they live in.
How inflammation rewires the colon
Dr. Emina Huang and her colleagues started with a hypothesis borrowed from other cancer types. Breast and pancreatic cancers had already shown a pattern: chronic inflammation causes scarring, which stiffens tissue, which in turn accelerates cancer growth. Could the same thing be happening in the colon of younger patients.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxTo test this, they examined tissue samples from colorectal cancer patients who'd had surgery. The results were striking. Both the tumors and the healthy tissue surrounding them were noticeably stiffer in patients diagnosed before age 50 compared to those diagnosed after. More importantly, the collagen—the protein that gives tissue its structure—was denser, longer, and more organized in the younger patients' samples. It looked like scarring had already remodeled the tissue before cancer even took hold.
When the researchers looked at gene activity, they found heightened expression of genes tied to collagen buildup, inflammation, and blood vessel formation in early-onset samples. The pattern was clear: chronic inflammation had been quietly reshaping the colon's physical landscape.
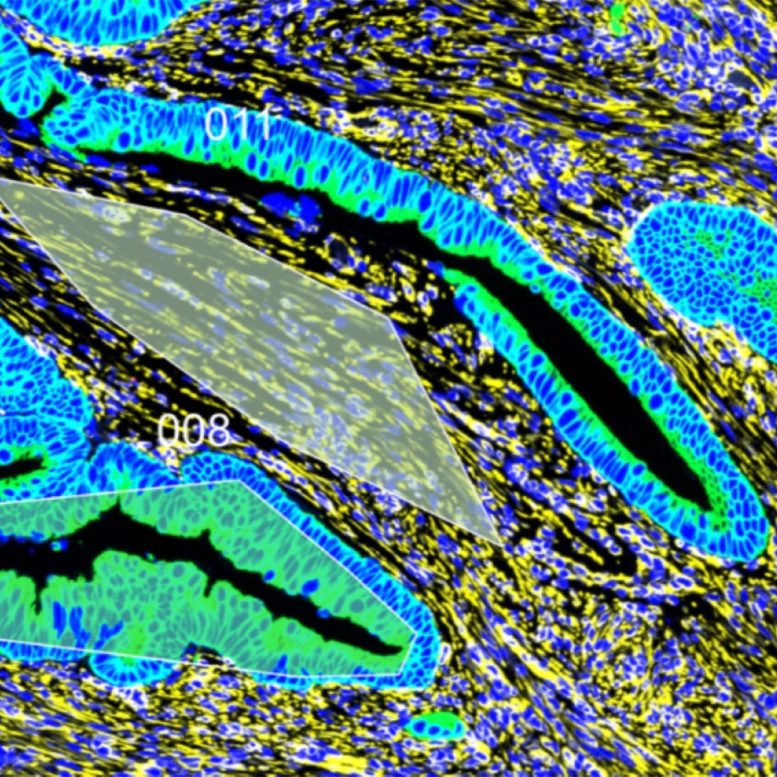
Stiffness becomes a growth accelerant
But tissue stiffness alone doesn't cause cancer. The researchers needed to show that a stiffer environment actually helps cancer cells thrive. They grew colorectal cancer cells on substrates with varying degrees of rigidity. The results were consistent: cells multiplied faster on stiffer surfaces. When they built three-dimensional organoid models—miniature tissue structures grown from cancer cells—the same pattern held. Stiffness accelerated growth.
This matters because it suggests the tissue environment isn't just a passive backdrop to cancer development. It's an active participant. A stiffer colon doesn't just tolerate cancer cells—it seems to encourage them.

What comes next
The findings open two practical doors. First, they suggest that disrupting the molecular pathways that sense and respond to tissue stiffness could slow or stop cancer growth—a strategy already being tested in other cancer types. Second, they hint at a diagnostic opportunity: if researchers can develop tests that measure intestinal stiffness, they could potentially identify younger people at risk before cancer develops, much like colonoscopies have done for older populations.
The inflammation that stiffens the colon typically builds over years, driven by diet, obesity, environmental factors, and other chronic conditions. Understanding this pathway doesn't eliminate those risk factors, but it gives researchers a new angle of attack—and patients a clearer reason to address inflammation before it reshapes their tissues.