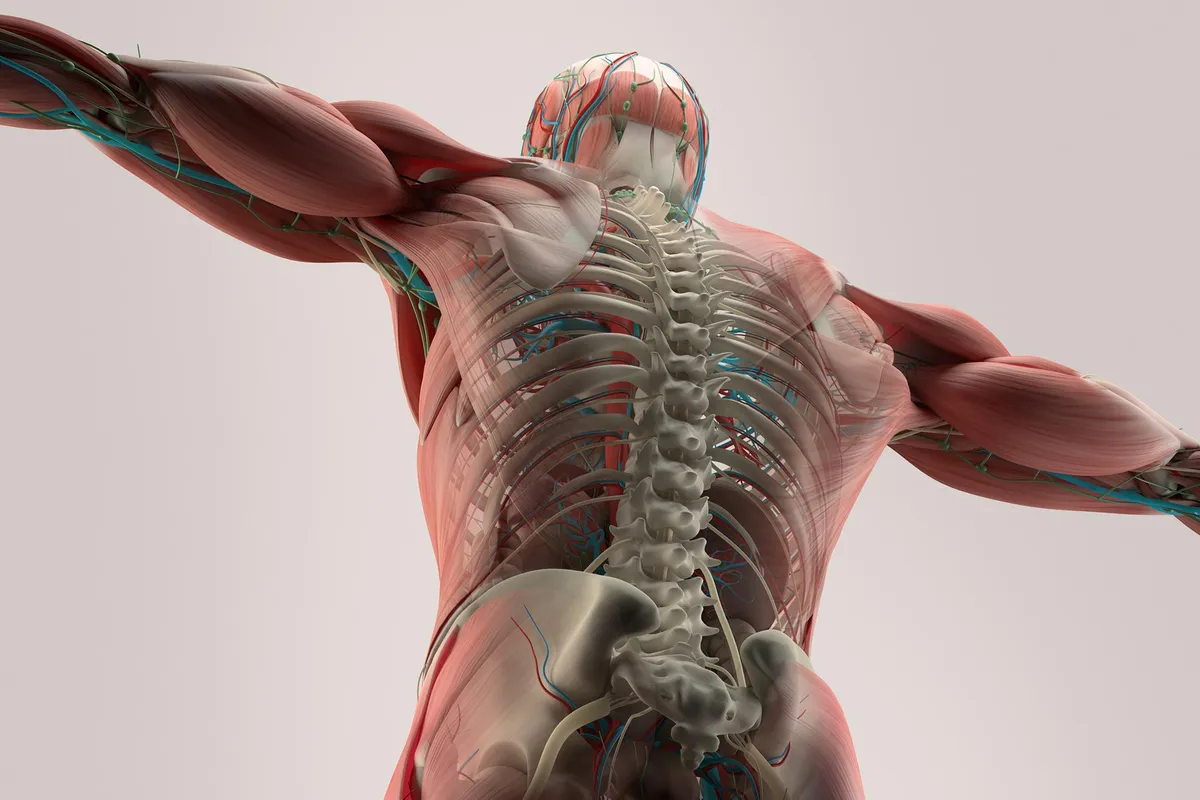
Researchers at Lund University have created something counterintuitive: a bone repair implant that works better because it contains no living cells.
The approach sidesteps one of transplant medicine's oldest problems. When surgeons graft living tissue from one person to another, the immune system often treats it as an invader. But this cartilage scaffold—stripped of its cells but still carrying the biological instructions cells need—seems to slip past those defenses. In animal testing, it triggered minimal immune reaction while guiding the body's own repair machinery to rebuild bone.
For the roughly two million people worldwide who need bone tissue transplants each year, this matters. Current options are often patient-specific, meaning surgeons harvest bone or cells from the person being treated. That adds procedures, cost, and unpredictability. A 65-year-old recovering from cancer surgery waits weeks for their own tissue to be processed. A younger patient with severe arthritis might need multiple procedures. Results vary person to person.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxHow a Dead Scaffold Still Talks to Living Tissue
The Lund team grew cartilage in the lab, then removed all the cells through a process called decellularisation. What remains is the extracellular matrix—the structural scaffolding that cells naturally build around themselves. But this isn't just empty framework. The matrix still holds growth factors, the biochemical signals that tell your body "rebuild here." When surgeons implant it, those signals activate your own cells to migrate in, colonize the scaffold, and gradually transform it into new bone.
It's less like a transplant and more like leaving behind an instruction manual your body already knows how to read.
"We show that it is possible to create a ready-made, so-called 'off-the-shelf' graft that interacts with the immune system and can repair large bone defects," says Paul Bourgine, who led the study. Because the material can be manufactured in advance and stored, hospitals could eventually stock these implants like any other medical device—no waiting, no customization needed.
What Comes Next
The team is now planning human trials, starting with severe defects in the long bones of arms and legs. They're also scaling up the manufacturing process to ensure the same quality and safety in production batches. The path from promising animal data to clinical approval typically takes years, but the foundation is there: a standardized, reproducible method that works with the body rather than against it.
For someone facing major bone loss, that's the kind of shift that changes what recovery looks like.