For eight decades, chemists have struggled with a molecular puzzle that kept them from reliably controlling how medicines are built. Now researchers at the University of St Andrews have found the missing piece — and it could reshape pharmaceutical manufacturing.
The problem sits at the heart of how drugs are made. Many molecules exist in two mirror-image forms, like left and right hands. For most medicines, only one of these "handed" versions actually works. The other can be ineffective or cause unwanted side effects. Getting the right hand to form reliably has been the bottleneck.
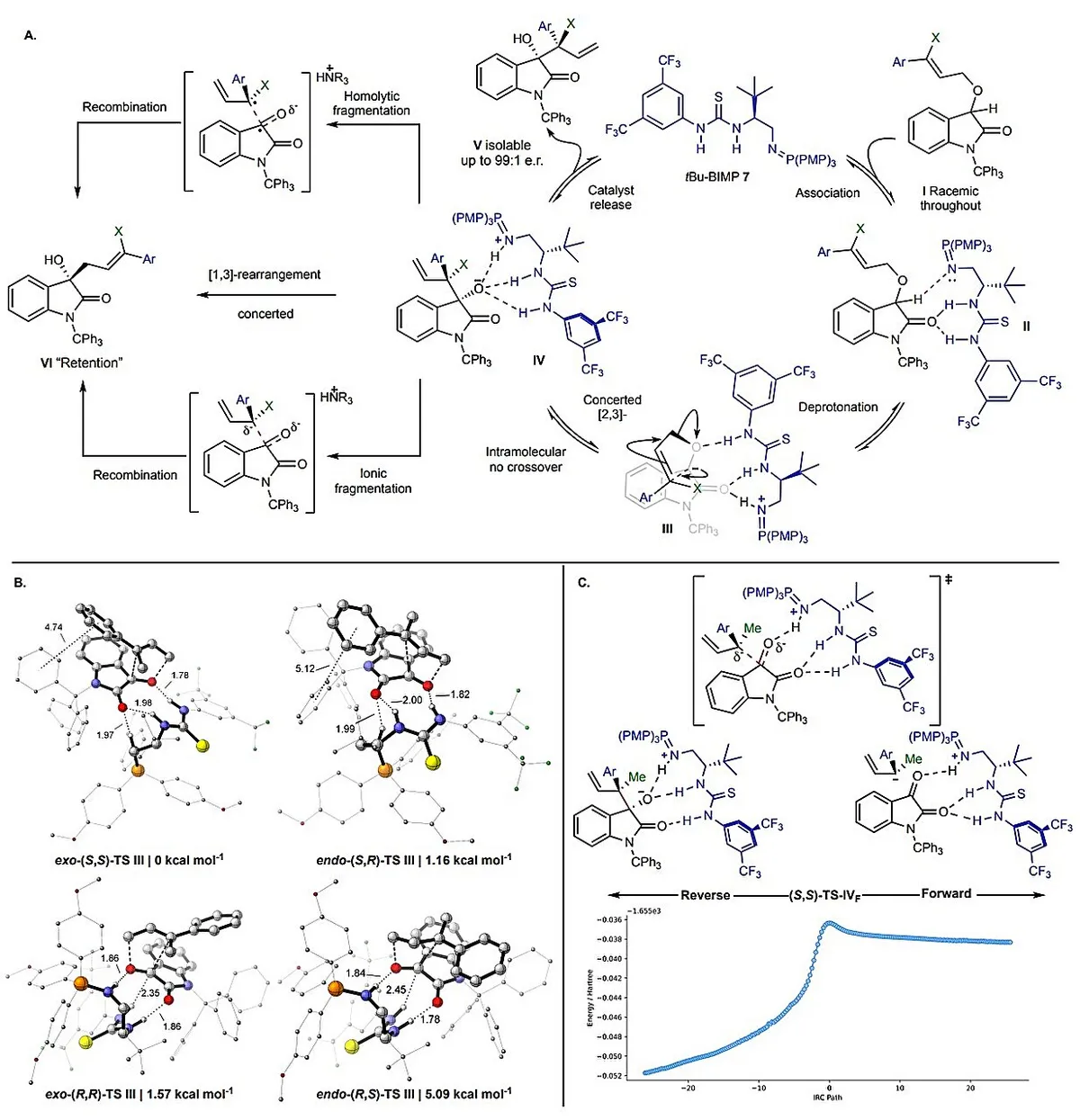
Enter the [1,2]-Wittig rearrangement — a chemical process discovered over 80 years ago that can reorganize atoms within a molecule. The trouble: it was wildly unpredictable. Chemists knew it could work, but controlling which handed form you'd end up with felt impossible. So they mostly avoided it.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxThe St Andrews team, working with colleagues at the University of Bath, finally cracked why. Using lab experiments paired with quantum chemistry calculations, they discovered that a catalyst guides the molecule through two distinct steps. First, it steers an initial asymmetric rearrangement that sets the molecule's handedness. Then comes the breakthrough: a previously hidden molecular "reshuffle" that locks that handedness in place.
"This discovery represents a fundamental shift in how we understand and control stereochemistry in rearrangement reactions," said Professor Andrew Smith, the study's lead author. The findings, published in Nature Chemistry, show that pathways chemists had written off as inaccessible are actually navigable — you just need to know how to read the map.
What does this mean in practice? Pharmaceutical companies could now use faster, cleaner, more selective methods to manufacture complex molecules of a single handedness. That translates to fewer wasted byproducts, shorter production timelines, and potentially lower drug costs. The same logic applies to advanced materials and specialty chemicals.
Dr. Matthew Grayson from Bath noted that the team's findings "open the door to new asymmetric transformations based on mechanistic pathways that chemists previously dismissed as inaccessible." In other words: entire categories of useful molecules that seemed out of reach are now within grasp.
This is the kind of foundational shift that doesn't make headlines but quietly changes what's possible in laboratories around the world.