More than 10,000 Americans are waiting for a liver transplant. Many will never get one. And even among those who might, some are too frail to survive the surgery itself. MIT engineers have just demonstrated something that could change that calculation: liver cells injected directly into the body, functioning as a backup organ without requiring a scalpel.
In a study published in Cell Biomaterials, researchers showed that injected hepatocytes—the liver's workhorse cells—remained alive and productive for at least two months, generating the enzymes and proteins a healthy liver produces. "We think of these as satellite livers," explains Sangeeta Bhatia, the lead researcher. "If we could deliver these cells into the body while leaving the sick organ in place, that would provide booster function."
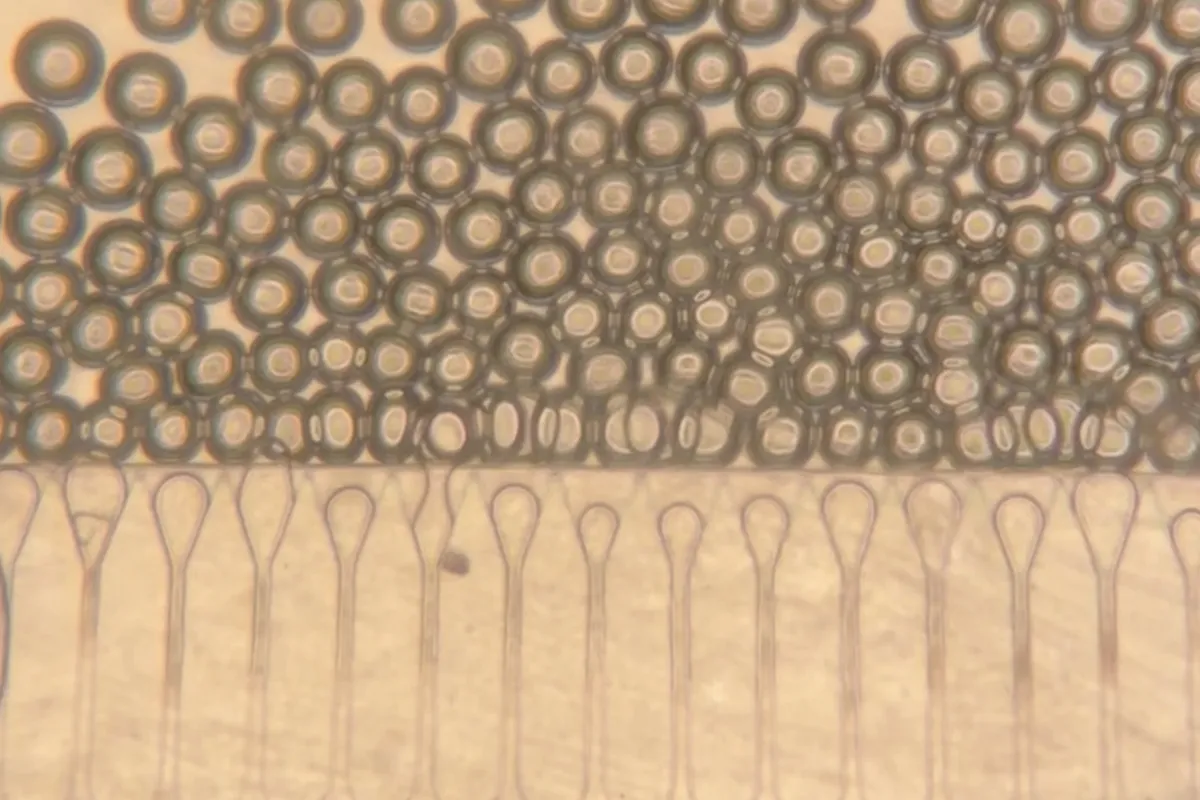
The human liver performs roughly 500 essential functions. It regulates blood clotting, filters bacteria from the bloodstream, metabolizes drugs. Most of this work falls to hepatocytes. The challenge has always been getting those cells to survive and integrate once transplanted. Bhatia's team cracked it by wrapping the cells in specially engineered hydrogel microspheres—tiny capsules that flow like liquid through a syringe, then solidify once inside the body. The mixture also includes fibroblasts, supportive cells that help hepatocytes survive and coax blood vessels to grow into the tissue.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxUsing ultrasound guidance, researchers injected this cell-sphere mixture into fatty tissue in the abdomens of mice. The results were clean: blood vessels grew directly into the graft. Nutrients flowed in. The hepatocytes stayed viable, secreting specialized proteins into the bloodstream for the full eight weeks of the study. "If the cells are injected without these spheres, they would not integrate efficiently," notes Vardhman Kumar, the study's lead author. "But these microspheres provide the hepatocytes with a niche where they can stay localized and become connected to the host circulation much faster."
What makes this approach genuinely different from traditional transplantation isn't just the lack of major surgery—though that matters enormously for patients too frail to survive it. It's the flexibility. If a patient needs another graft later, or a different therapy, the barriers are far lower than undergoing surgery again. It can also work as a bridge: supporting a patient's liver function while they wait for a donor organ, or indefinitely if no organ materializes.
The current version would require immunosuppressive drugs to prevent rejection, the same as traditional transplants. But researchers are already exploring ways to develop "stealthy" hepatocytes that could evade immune detection, or to use the hydrogel spheres themselves to deliver immunosuppressants directly to the graft site—potentially reducing the systemic side effects that come with drugs flooding the entire body.
The work is still early. Mouse studies aren't human trials. But the trajectory is clear: a therapy that could reach thousands of people currently trapped in the waiting-list limbo, and give doctors an option for patients who were never candidates for surgery in the first place.