Fruit fly larvae can regrow entire wings after lethal radiation exposure. For half a century, scientists watched this happen and had no idea why. Now researchers at the Weizmann Institute have cracked the code—and what they found suggests why some cancers come back stronger after treatment.
The discovery centers on caspases, enzymes best known for triggering cell death. But these enzymes do something unexpected: they can keep certain cells alive, even as they activate the self-destruct signal. Those cells then drive the regeneration of damaged tissue. The catch is that cancer cells may use the same trick to survive treatment and regrow more aggressively.
The cells that refuse to die
When fruit fly larvae were exposed to high-dose radiation in the 1970s, their epithelial tissues (the skin-like layers lining their organs) would surge back to life. New cells multiplied rapidly, restoring the damaged wing tissue within days. This process, called compensatory proliferation, happened in humans too—skin heals after burns, organs repair after injury—but the molecular machinery behind it remained a mystery.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxDr. Tslil Braun and his team used genetic tools to track what happened at the cellular level during radiation exposure. They found something surprising: a population of cells they named DARE cells (Damage-Activated Regeneration-Enabling cells) activated their self-destruct program but then stopped mid-process and survived anyway. These cells didn't just live—they multiplied rapidly and repaired nearly half the damaged tissue within 48 hours.
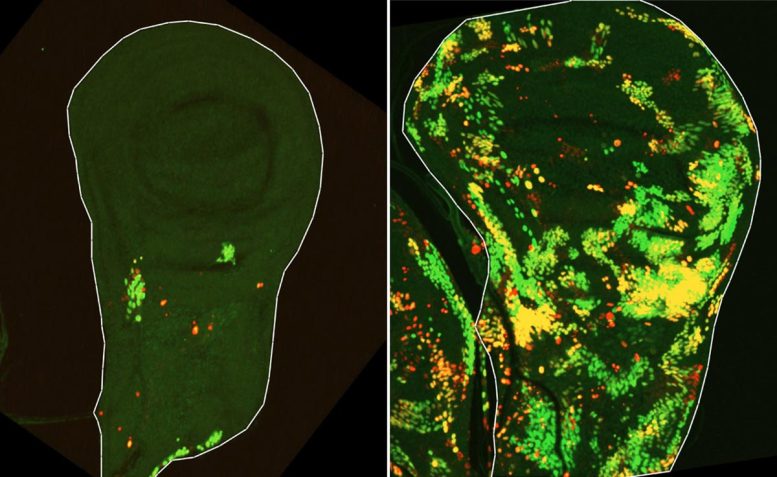
The researchers also discovered a second population, NARE cells, which survived without triggering the death signal at all. But here's what matters: when they removed DARE cells from the system, tissue regeneration stopped entirely. DARE cells were essential. Even more intriguingly, dying cells in the surrounding tissue sent signals that activated DARE cells, as if the damage itself was calling for help.
How a protein holds back the executioner
The real breakthrough came when the team figured out why DARE cells could start the death process and then pump the brakes. A molecular motor protein was tethering the initiator caspase to the cell membrane, preventing it from activating the "executioner" caspases that would normally finish the job. When researchers silenced this motor protein, DARE cells died and tissue regeneration failed.
That detail matters because the same motor protein is often overactive in cancer cells—a known driver of tumor growth. It's one mechanism cancer cells use to dodge apoptosis and survive treatment.
The inheritance problem
Here's where the cancer connection becomes sharper. When the researchers exposed the same tissue to radiation a second time, something ominous happened. The descendants of DARE cells were seven times more resistant to cell death than the original tissue. The tissue had become hardened, more resistant—exactly what oncologists see in recurrent tumors that regrow after radiation therapy and become more aggressive.
This suggests a mechanism for why some cancers come back worse: the cells that survive initial treatment become inherently more resistant, passing that resistance to their descendants.
A balancing act
The team also discovered that tissue repair isn't a free-for-all. DARE cells promote growth in NARE cells by secreting growth signals, but NARE cells send back inhibitory signals that prevent DARE cells from overgrowing. It's a negative-feedback loop—a built-in brake on regeneration. Without this balance, healing would turn into uncontrolled growth, which is essentially what cancer is.
Prof. Eli Arama, who led the research, notes that this knowledge opens new possibilities. Many cancers originate in epithelial cells that lose growth control, and traditional treatments try to force them into apoptosis. Understanding why that sometimes fails—and how cancer cells exploit the same survival mechanisms that healthy tissue uses to heal—could point toward better treatments. The findings also suggest ways to accelerate healing in healthy tissue after injury, without triggering the resistance that makes tumors dangerous.
The question now is whether these mechanisms work the same way in human tissue. If they do, the next generation of cancer treatments might target not just cell death, but the survival tricks that let cancer cells escape it.