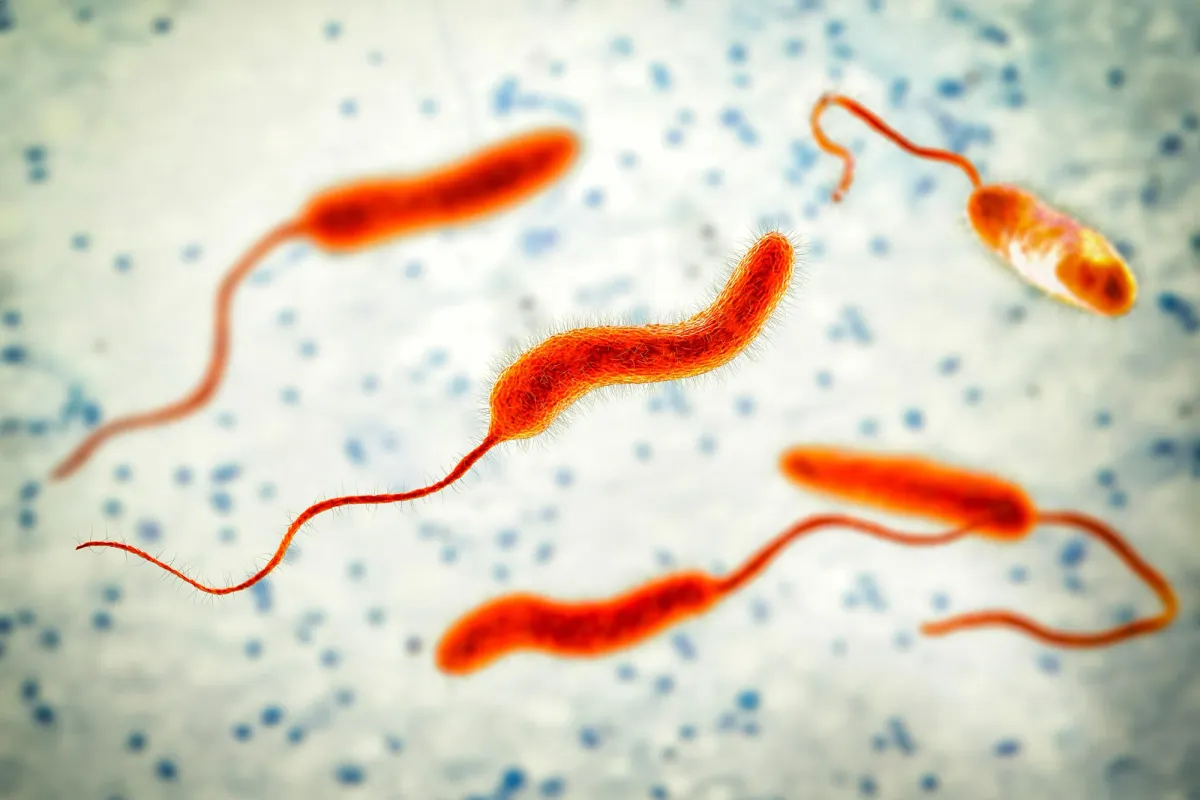
For decades, scientists thought they had cholera all figured out. A nasty bacterium, Vibrio cholerae, causes severe, often fatal, diarrheal disease. It spreads through contaminated water and food, and it’s been raging in its seventh global pandemic since 1961, sickening millions each year. The Ganges Delta? That was the undisputed global headquarters for this watery menace.
Turns out, cholera has a secret frenemy, and it's been fighting a silent war for 20 years. This battle affects everything: how sick people get, and how far the disease spreads. And it just might upend everything we thought we knew about stopping it.
The Unsung Hero: Bacteriophage ICP1
Researchers from the Wellcome Sanger Institute and icddr,b spent two decades meticulously tracking cholera in Bangladesh and North India. They discovered that Vibrio cholerae isn't just chilling; it's locked in an evolutionary arms race with a virus called bacteriophage ICP1. Phages are viruses that infect bacteria, essentially turning them into viral factories. They’re harmless to humans but absolute nightmares for bacteria.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxWhat's wild is the trade-off: when cholera bacteria gain defenses against ICP1, they become less virulent and less likely to spread far. It’s like putting on heavy armor that slows you down. But when they shed those defenses, they become super-spreaders, causing more severe illness. The phage, meanwhile, is constantly evolving new ways to bypass those defenses. It's an ongoing, microscopic tug-of-war, and we're just now understanding its implications.
Rethinking Cholera's Global Game
This 20-year deep dive into over 2,300 cholera samples created the most detailed dataset for the region, and it basically rewrites the cholera playbook. First, the Ganges Delta isn't the sole global source; it's the broader Ganges Basin. More importantly, cholera doesn't just flow with the rivers. It tends to respect national borders, suggesting human movement and population density are far bigger drivers of its spread than water currents. Which, if you think about it, makes a lot of sense for a disease that relies on people sharing things.
Knowing about this constant bacterial-viral battle opens up some fascinating possibilities. We could develop early warning systems to spot those "unarmored", highly dangerous cholera strains before they go wide. Imagine tracking the bacteria's genetic defenses in real-time, knowing exactly when a super-spreader is about to emerge. Health officials could then deploy vaccines and sanitation efforts with surgical precision.
Even cooler, the phages themselves could become a new treatment. Using ICP1 to directly fight cholera infections in humans? That’s like turning the bacteria’s own natural predator into medicine. It’s a beautifully elegant solution, if we can pull it off.
This study doesn't just update our maps; it gives us a whole new ecological lens to view cholera. By understanding the intricate dance between bacteria and virus, we might just be able to outsmart one of humanity's oldest and most persistent foes.