For the first time, scientists have watched atoms rearrange themselves in the trillionths of a second before X-ray radiation triggers their decay — and what they found changes how we understand radiation damage at the most fundamental level.
When X-rays hit matter, they leave atoms in unstable, excited states. These atoms then relax through decay processes that can snap chemical bonds and harm the biomolecules cells depend on. The question that's haunted researchers: what exactly happens in those first fractions of a second, and can we predict or prevent the damage that follows.
An international team led by the Fritz Haber Institute in Berlin focused on a specific decay mechanism called electron-transfer-mediated decay (ETMD). It's particularly important because it produces low-energy electrons through interactions between neighboring atoms — the kind of electrons that cause the most chemical havoc in water and living tissue. Understanding ETMD means understanding a major pathway for how radiation harms biology.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxWatching atoms move in real time
To catch atoms in the act, the researchers built a deliberately simple system: one neon atom weakly bound to two krypton atoms. They hit the neon with soft X-rays, then tracked what happened for up to one picosecond — an eternity on the atomic scale — using an advanced reaction microscope at synchrotron facilities in Berlin and Hamburg. They also ran thousands of computer simulations to map every possible path the atoms might take.
What emerged was unexpected. The atoms didn't stay put. Instead, they roamed across the molecular landscape, continuously reshaping their arrangement and directly steering when and how decay occurred. "We can literally watch how the atoms move before the decay happens," says Florian Trinter, one of the lead researchers. "The decay is not just an electronic process — it is steered by nuclear motion in a very direct and intuitive way."
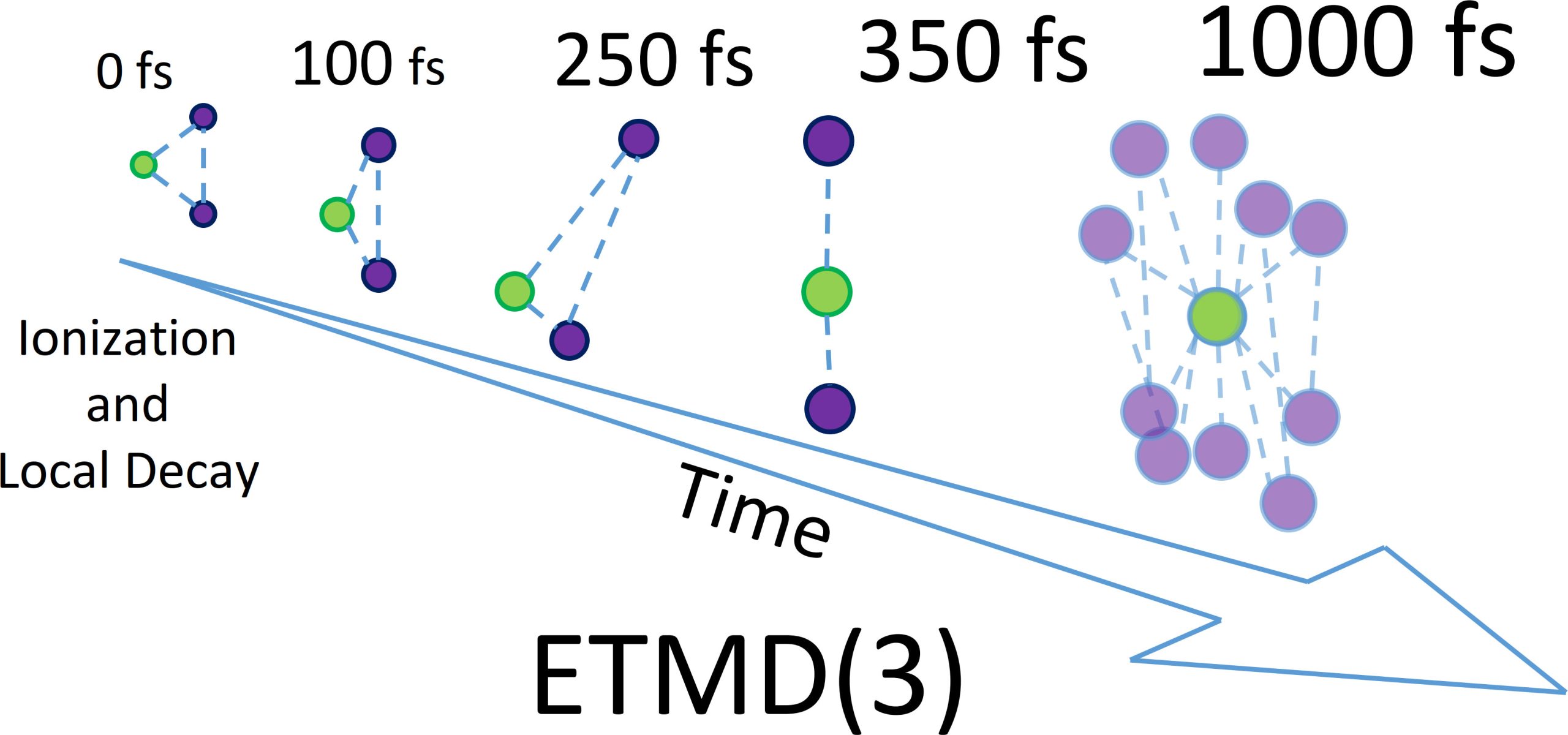
The team discovered that decay doesn't happen from a single frozen structure. Early on, it occurs near the atoms' resting position. But as time passes, one krypton atom drifts closer to the neon while the other pulls away — the perfect geometry for electron transfer and energy release. Later, the system explores almost linear and wildly distorted shapes, with atoms swinging back and forth in a pendular motion. These shifting geometries cause decay rates to swing by nearly tenfold depending on the moment.
"The atoms explore large regions of configuration space before the decay finally takes place," explains Till Jahnke, the study's senior author. "This shows that nuclear motion is not a minor correction — it fundamentally controls the efficiency of non-local electronic decay."
Why this matters for radiation protection
This level of detail matters because ETMD efficiently produces exactly the kind of low-energy electrons that cause chemical damage in cells and water. By understanding how molecular motion shapes this decay, researchers can now build better models of radiation damage in biological environments — and eventually, better ways to predict or limit it. The findings also provide a benchmark for developing theoretical approaches that can scale up from three atoms to the complexity of actual cells and tissues.
The work opens a new door: using electronic decay itself as a probe to image ultrafast molecular motion with unprecedented precision. What happens next is researchers extending these insights from this simple three-atom system to liquids, solvated ions, and eventually to the messy, crowded reality of living matter.









