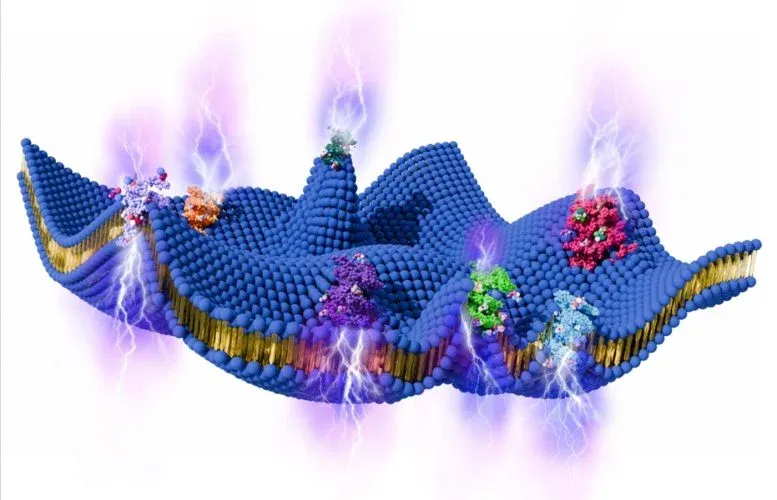
Your cells might be running on a power source you never knew existed. Scientists have discovered that the thin membrane surrounding every cell—constantly flexing and rippling from the activity inside—generates real electrical signals strong enough to rival nerve impulses in your brain.
It's the kind of finding that makes you rethink how cells actually work. We tend to think of cell membranes as passive barriers, but they're anything but still. Proteins are shifting shape, chemical reactions are firing, and the cell is breaking down ATP molecules to power itself. All that internal commotion pushes and pulls on the membrane, creating tiny waves and bends at the molecular level. According to a new theoretical framework, these movements aren't just mechanical noise—they're generating voltage.
The mechanism is called flexoelectricity, a physical effect where deformation in a material creates electrical charge. When the membrane bends, it produces a voltage difference across itself. The researchers found these voltages can reach up to 90 millivolts—the same ballpark as the electrical signals neurons fire when sending messages through your brain. Even more striking, the timing matches: these voltage fluctuations happen over milliseconds, mirroring the speed of actual nerve signals.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxHow Cells Move Ions Against the Flow
What makes this particularly interesting is what comes next. The electrical signals created by membrane motion could actively transport ions—the charged particles that are central to how cells communicate and maintain their internal balance. Normally, ions flow naturally from areas of high concentration to low concentration, like water flowing downhill. But the model suggests that active membrane fluctuations could push ions in the opposite direction, working against that gradient.
Whether ions move forward or backward, and how strongly, depends on two properties of the membrane: how easily it bends (elastic properties) and how it responds to electrical fields (dielectric properties). These two factors work together to determine the direction and strength of ion transport. It's a neat bit of physics—the same membrane motion that generates the voltage also uses that voltage to move ions where they need to go.
The implications ripple outward. If individual cells generate electricity this way, what happens when millions of cells coordinate their membrane activity? The researchers suggest this same framework could explain how tissues produce collective electrical behavior—potentially shedding light on how we sense the world, how our neurons fire, and even how living systems harvest energy at the cellular level.
There's also a practical angle: understanding how cells generate and control electrical signals through pure mechanics could inspire new materials that mimic this behavior—smart materials that respond to their environment without traditional electronics. The bridge between biology and engineering just got a little clearer.