You know oxygen. You breathe it, you need it. But just below it on the periodic table? A whole family of elements called chalcogens, and they've been quietly running a crucial show inside your cells.
Sulfur is the most famous sibling, a workhorse in managing your cells' redox balance — essentially, the cellular equivalent of keeping your house from going up in flames. Too much oxidation and you've got oxidative stress, which sounds exactly as fun as it is: linked to aging and a whole host of diseases. Sulfur-packed molecules like glutathione are your internal firefighters, battling harmful molecules to keep things stable.
But sulfur's heavier relatives, selenium and tellurium, have been a bit more mysterious. Researchers knew they were involved in these redox processes, but studying the unstable molecular chains where these different chalcogens link up? That's been a tough nut to crack. Mostly because the usual methods, like mass spectrometry, couldn't actually see the connections.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxUnmasking the Chalcogen Clan
Enter Kyoto University. A team led by Kazuma Murakami decided it was time to get a closer look. They'd been intrigued for years by how tiny atomic tweaks could have big biological ripple effects. Chalcogen chemistry, they realized, was an untapped goldmine for understanding redox biology.
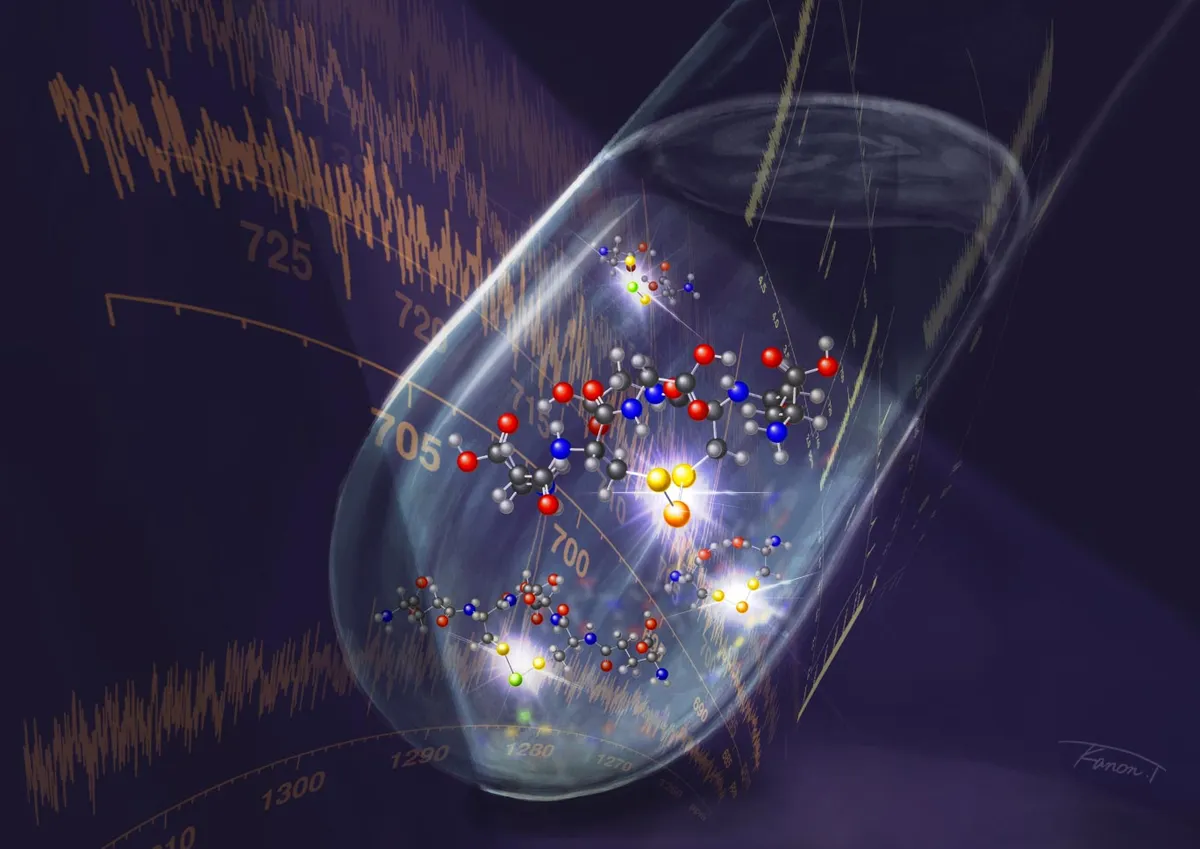
So, they cooked up a new method. They took oxidized glutathione-cystine molecules, added some selenium or tellurium atoms, and watched the magic happen in water. The real secret sauce? Using something called 1H-detected 77Se/125Te nuclear magnetic resonance spectroscopy (NMR). Which, if you think about it, is both impressive and slightly terrifying in its specificity.
This allowed them to directly observe these unstable bonds between different chalcogen atoms, creating what they called "heterologous trichalcogenide molecules." And what did they find? These compounds are absolute powerhouses in redox reactions, far more active than previously understood.
Murakami put it plainly: this is the first time anyone has directly seen these heterochalcogen bonds in action within redox systems using spectroscopy. By combining multinuclear NMR with superchalcogenide chemistry, they've basically given scientists a brand new set of glasses to look at life's tiniest, most crucial battles.
The Health Implications? Huge.
This isn't just a win for chemistry nerds (though they're probably having a field day). This new technique means we can start designing new redox-active molecules with far greater precision. Think new functional biomolecules, peptides, and a deeper understanding of oxidative stress and diseases like ferroptosis (a particularly nasty type of cell death).
Next up, the team plans to apply their new superpower to even more complex biomolecules, continuing to unravel the biological roles of these chalcogen-modified glutathione derivatives. The ultimate goal? New redox-active compounds for future medical treatments. Because who doesn't want better internal firefighters?