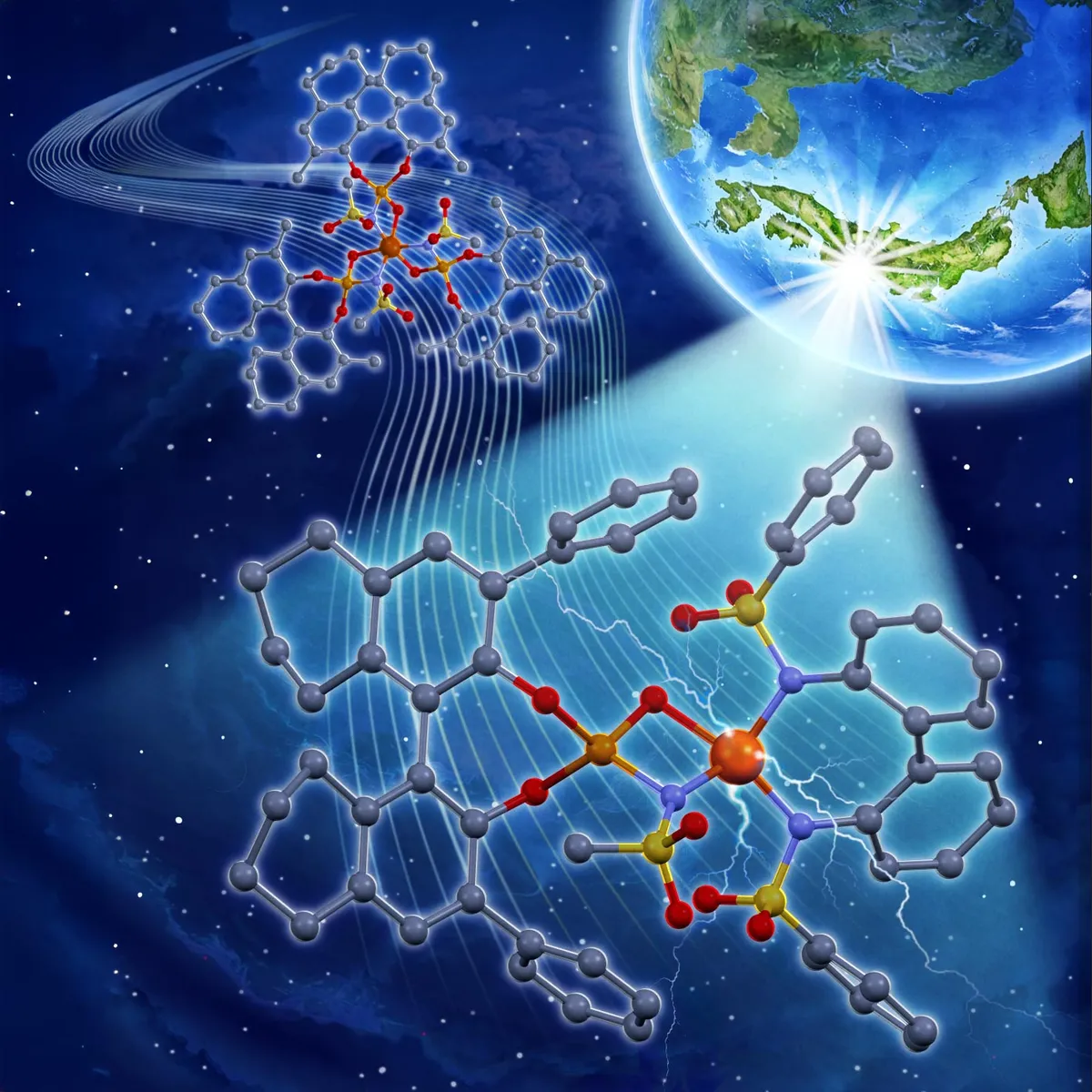
A team at Nagoya University just cracked something chemists have wanted for years: a way to build complex drug molecules using cheap, abundant iron instead of rare metals — and blue LED light instead of expensive energy sources.
The breakthrough hinges on how photocatalysts work. These materials trigger chemical reactions when light hits them, and in drug manufacturing, they're invaluable for building molecules with precise three-dimensional shapes. The problem has always been the metals themselves. Ruthenium and iridium are excellent at this job, but they're rare and costly. Iron works too, but earlier versions needed massive amounts of expensive ligands — molecular guides that steer the shape of the final product.
The new design, published in the Journal of the American Chemical Society, cuts ligand use by two-thirds. Here's how: instead of loading the iron atom with three chiral ligands (only one of which actually did the real work), the team paired one inexpensive, structurally neutral ligand with a single chiral ligand. The neutral one boosts the catalyst's overall power. The chiral one controls the shape. Same result, far less waste.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxBuilding molecules that matter
Under blue LED light, this iron catalyst can now link two molecular pieces into a six-membered ring — a structure that appears constantly in nature and in medicines. The team proved it works by synthesizing (+)-heitziamide A, a compound from medicinal plants that suppresses immune inflammation. This was the first time anyone had made the naturally occurring version asymmetrically (meaning they could control which of its two mirror-image forms they created).
That matters because enantiomers — those mirror-image versions — behave completely differently in the body. One might be medicine. The other might be useless or harmful. Being able to selectively build one or the other is a huge deal in drug development.
What makes this genuinely significant is the economics. Rare metals are scarce by definition. Blue LEDs are cheap and getting cheaper. Iron is everywhere. If this approach scales, it doesn't just mean lower costs for manufacturers — it means drug synthesis becomes more accessible to labs in countries that can't afford rare-metal catalysts. It's a small shift in the chemistry toolkit that could ripple outward.
The researchers are already working on asymmetric syntheses of other bioactive compounds using the same method, suggesting this isn't a one-off win but the foundation of a broader toolkit.