Scientists have long puzzled over how the first living cells emerged from non-living chemistry. The leading theories point to warm hydrothermal vents or drying desert pools — places where simple molecules could concentrate and react. But new experiments from Tokyo Tech suggest we've been looking in the wrong climate. Life might have started in the cold.
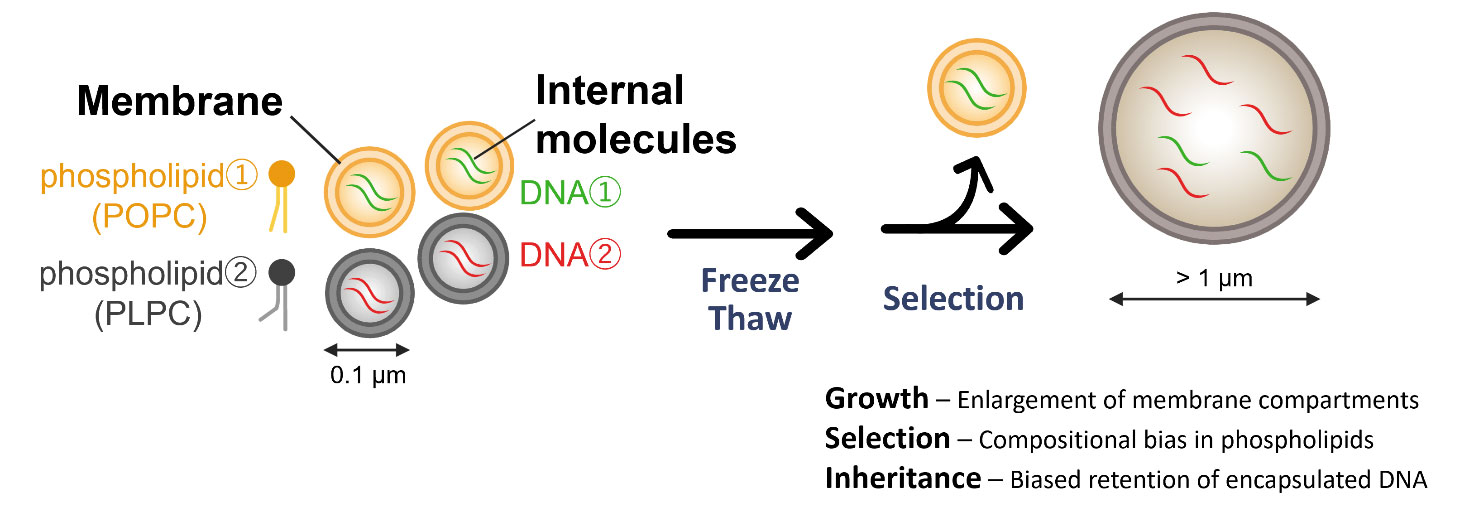
Researchers created tiny bubble-like structures called vesicles using lipids similar to those in modern cell membranes, then subjected them to repeated freeze-thaw cycles. What they found was striking: when ice formed and melted, something unexpected happened. Vesicles made from certain types of lipids — those with more unsaturated bonds — began fusing together into larger compartments. More importantly, they held onto DNA trapped inside them far better than their saturated counterparts.
How freezing could have jumpstarted life
The mechanism is elegant. When water freezes, it concentrates whatever molecules are dissolved in it into smaller and smaller pockets of liquid. Imagine a puddle on early Earth, full of organic molecules and primitive lipid bubbles. As temperatures drop, the remaining liquid becomes increasingly crowded. Vesicles are forced closer together and begin to merge. When they fuse, their contents mix — and that mixing is where chemistry becomes interesting.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxThe researchers tested this by comparing two types of lipid membranes. Vesicles made from POPC (a more saturated lipid) stayed separate and rigid through freeze-thaw cycles. But those made from PLPC (more unsaturated) fused together readily and retained more of the genetic material trapped inside. This matters because the earliest cells would have needed to both grow and preserve whatever genetic information they contained. Freezing provided both.
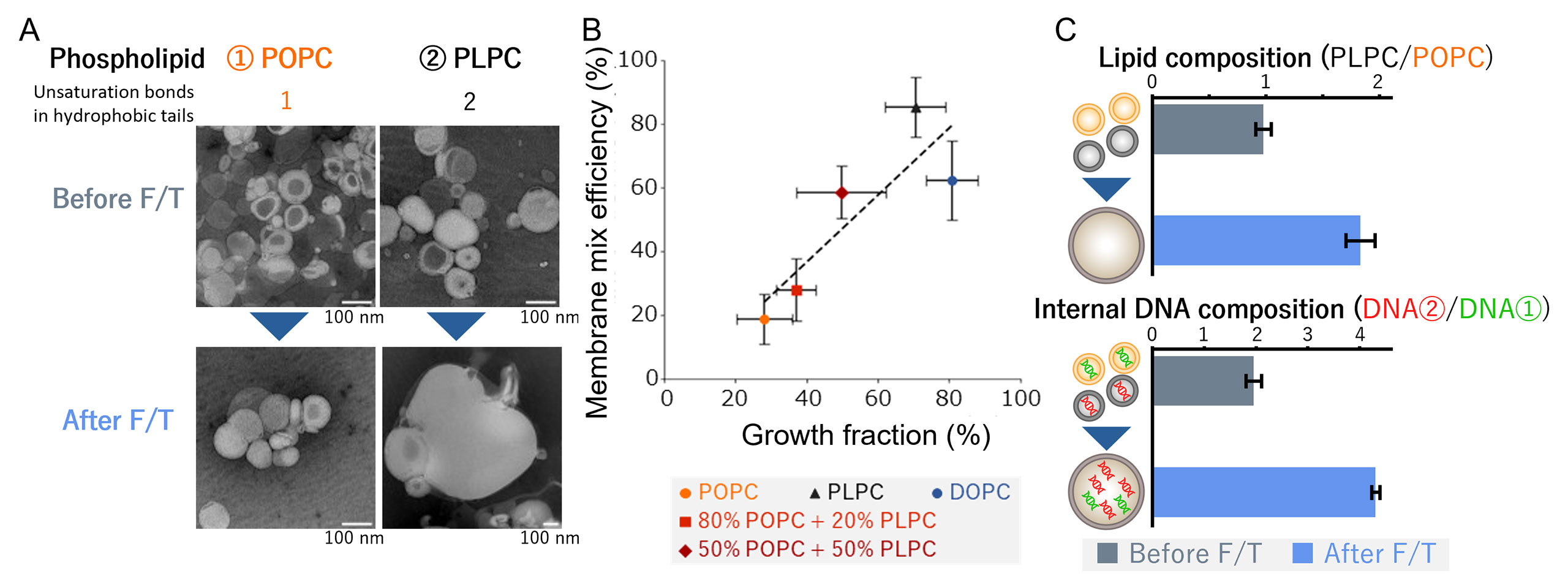
Why does this shift our thinking. Most origin-of-life research has focused on warm environments — hot springs, tidal pools, mineral-rich vents. These make intuitive sense; chemistry moves faster when it's warm. But early Earth had plenty of frozen regions too, perhaps especially in the first few hundred million years. And cold has a hidden advantage: it's a natural concentrator. Freeze-thaw cycles don't just mix molecules; they select for membranes that can survive the process.
The study suggests that repeated cycles over thousands of years could have progressively refined protocells — selecting for membrane compositions that fused efficiently, retained genetic material, and allowed chemical reactions inside. Eventually, as molecular complexity increased, the systems inside these bubbles might have developed the ability to replicate themselves. That's when Darwinian evolution could begin, and life as we know it could emerge.
This doesn't mean warm-water scenarios are wrong. Early Earth probably had many environments where chemistry was happening. But it does expand where we should look when hunting for clues about life's origins — and it reminds us that harsh conditions, even freezing ones, can be surprisingly creative.










