Every year, over 100 billion nitrile rubber gloves end up in landfills or incinerators—a waste stream so massive it's almost invisible. Most of these gloves are used once in hospitals, clinics, and care settings, then burned. That incineration releases the very thing we're trying to remove from the atmosphere: carbon dioxide.
But researchers at Aarhus University in Denmark have found a way to flip this equation. They've figured out how to take discarded rubber gloves and turn them into material that captures CO2 instead of releasing it.
"In our experiments, we converted the glove so that it can capture CO2 instead of becoming a waste product that releases CO2 and other harmful gases during incineration," explains Simon Kildahl, a postdoctoral researcher in the chemistry department who led the work.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxThe breakthrough sounds almost too neat—waste material solving a climate problem rather than creating one. But it addresses a real recycling gap. Unlike plastic bottles, which flow through deposit-return systems, rubber materials are nearly impossible to recycle into their original form. They get burned. End of story. Kildahl's team, part of the Novo Nordisk Foundation CO2 Research Center at Aarhus, asked: what if we stopped trying to recycle them the old way and used them for something else entirely.
From shredded gloves to CO2 traps
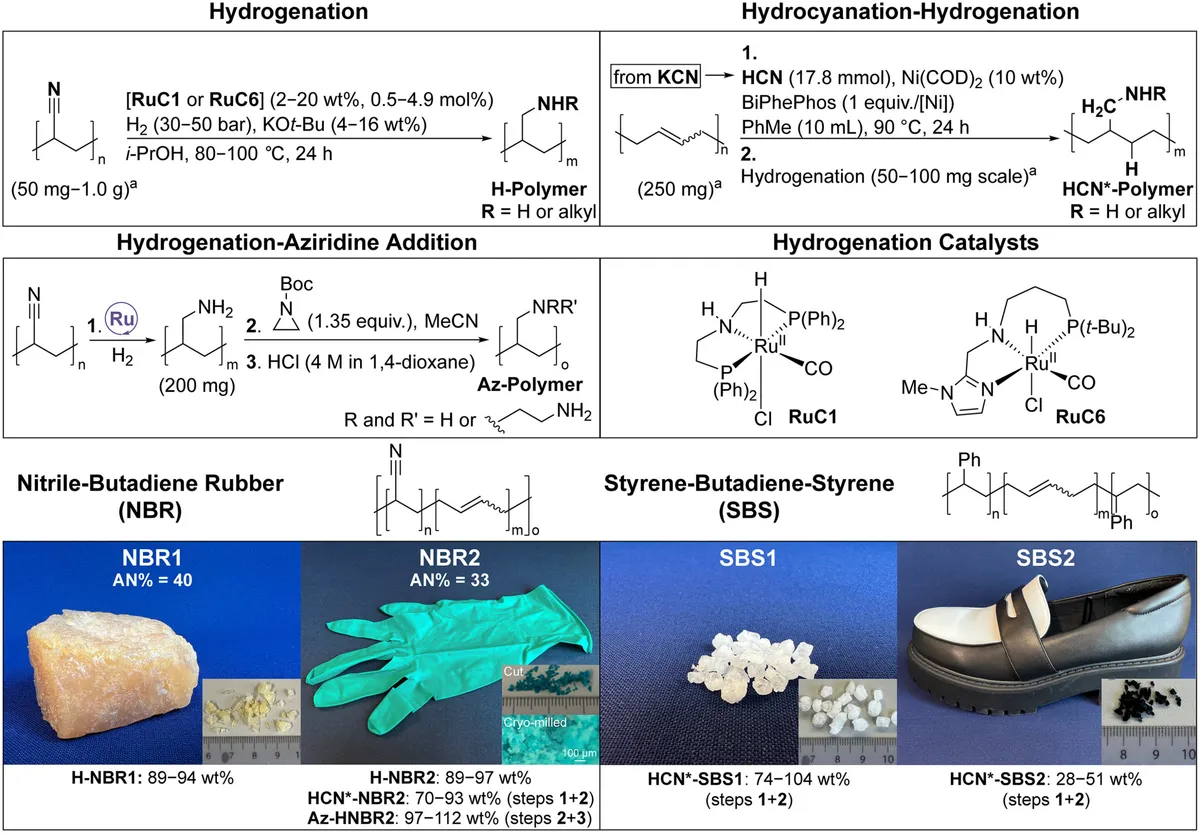
The process itself is elegant. Rubber gloves are shredded into small pieces, then exposed to a ruthenium-based catalyst and hydrogen gas. This chemical transformation restructures the material so it can capture CO2 from flue gas—the exhaust streams from power plants and industrial facilities. When the material is heated later, it releases the captured CO2, which can either be stored underground or converted into other products through Power-to-X technology. Then the material is ready to capture more CO2. A continuous cycle.
This approach has a climate advantage that matters. The UN's climate panel estimates we need to remove between 5 and 16 billion tons of CO2 from the atmosphere annually by 2050. Most current CO2 capture materials require new oil extraction to produce them—which undermines the whole climate benefit. Using waste gloves sidesteps that problem entirely.
"That is why it is smart to utilize a waste material available in such large quantities, rather than extracting more oil from the ground," Kildahl says. "With the rubber glove, we can create a CO2 capture material where almost every atom in the product comes from waste, except for a small amount of hydrogen, which can ideally be obtained from water."
The work isn't ready for commercial deployment yet. It's currently at laboratory scale—measured in grams—and sits at level three or four on the Technology Readiness Level scale, which runs from early idea to fully commercialized. Scaling chemical reactions from grams to kilograms introduces unexpected complications. Reactions behave differently at larger volumes, and early results, while promising, still need refinement.
Two concrete obstacles remain. The ruthenium-based catalyst is expensive, making the process economically uncompetitive right now. And the material's CO2 capture performance needs improvement before it can compete with existing methods. But Kildahl is genuinely optimistic about the timeline. "We have reached a proof of concept. It is entirely possible that we can reach level five or six in the near future if we can improve the scalability and the economy of the reaction."
That's the stage where this moves from laboratory curiosity to something that could actually work at scale. With 100 billion gloves produced every year, there's no shortage of raw material waiting.