How Evolution Moved Into the Lab
Evolution has always been nature's best engineer. Inside cells, countless variations of proteins emerge and disappear—and the ones that work best stick around. Humans figured this out millennia ago: early farmers picked the strongest crops and healthiest animals to breed, letting evolution do the heavy lifting.
Modern scientists do something similar in laboratories. They use a technique called directed evolution to redesign proteins that fight disease, clean clothes, or manufacture chemicals. The process works: researchers have engineered enzymes and antibodies that outperform their natural versions.
But there's a catch. Standard directed evolution methods favor proteins that stay "on" all the time. They reward constant activity. In real biology, though, most proteins don't work that way. A protein might need to flip on, then off, then on again—like a light switch, or a logic gate that says "yes" only when two conditions are met simultaneously. When evolution only rewards the "always on" state, the protein loses the ability to switch properly. The result: a broken protein.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxThis limitation has made it nearly impossible to evolve proteins with complex, dynamic behavior using traditional methods.
Light as a Tool for Better Evolution
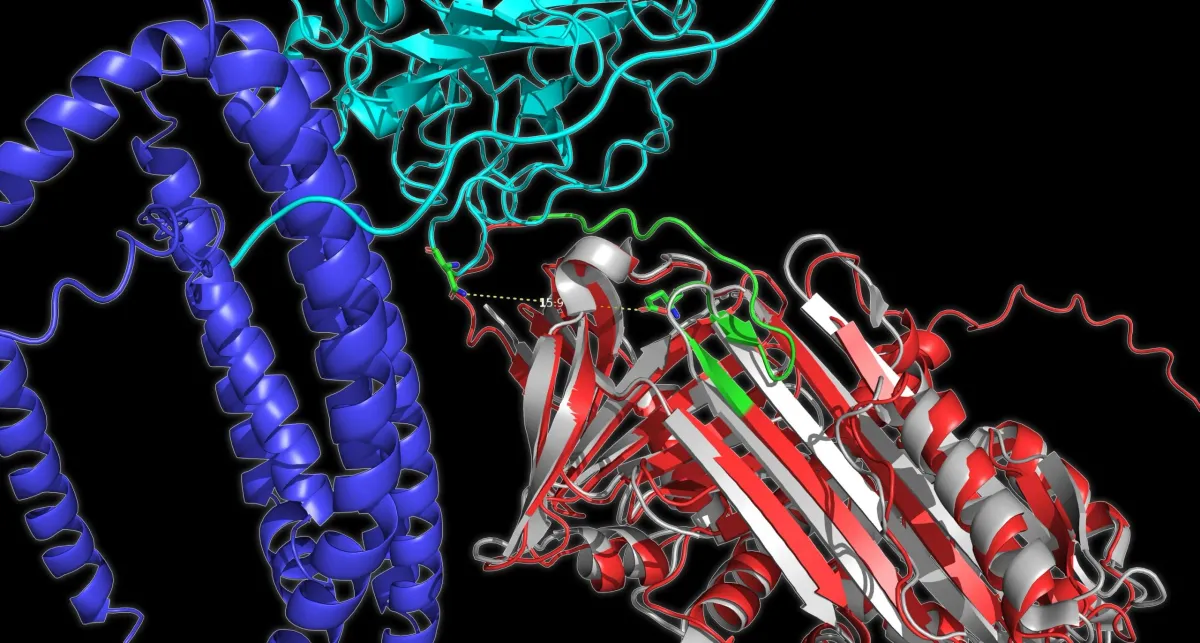
Researchers at EPFL's Laboratory of the Physics of Biological Systems, led by Sahand Jamal Rahi, just solved this problem with an approach called "optovolution." The idea is elegant: use light to guide evolution toward proteins that can switch states, sense their environment, and even perform simple computational tasks.
The team started with budding yeast, the same organism that ferments beer. They rewired the yeast cell cycle so that survival depended entirely on how well the target protein could switch on and off. A protein that stayed active too long? The cell would stall and die. One that couldn't activate at all? Dead too. Only cells with proteins that switched at exactly the right moments kept dividing.
Then came the clever part. The researchers used optogenetics—a technique that controls genes with light—to deliver precisely timed light pulses to the yeast. Each cell cycle takes about 90 minutes, so the system ran thousands of pass-or-fail tests per day. Proteins that switched well allowed their cells to survive and reproduce. Bad switchers were eliminated. No manual screening needed. No guesswork.
What Proteins Can Now Do
Using this method, the team evolved several different protein types. They improved a light-controlled transcription factor and generated 19 new variants that respond to different colors of light—including green, which researchers had always struggled to engineer. They also evolved a red light system that no longer requires adding chemicals to the yeast; instead, the protein learned to use light-sensitive molecules already present in the cell.
Most striking: they evolved a transcription factor that works like a single-protein computer. It activates genes only when two different inputs arrive at the same time—one light signal and one chemical signal. It's a biological AND gate.
For context, proteins that can sense, switch, and compute are fundamental to how cells respond to their environment, make decisions, and divide. Getting evolution to favor these behaviors—rather than just raw activity—opens new doors. Scientists could now design smarter cellular circuits, create optogenetic tools that respond to multiple colors independently, or understand how complex protein behaviors emerge naturally over time.
The technique is still young, but the implications are quietly remarkable. By letting evolution happen inside living cells—guided by light rather than human hands—researchers have moved closer to how nature actually engineers biology.