A team at the University of Oxford has figured out how to see something that's been invisible to researchers for years: the binders that hold lithium-ion battery electrodes together. It sounds like a small thing — these binders make up less than 5% of an electrode's weight — but they control whether your phone charges quickly, how long the battery lasts, and whether the whole thing stays structurally sound.
The problem was that binders are essentially invisible. They lack distinctive features under standard imaging, and their tiny presence made them nearly impossible to track inside an electrode. So researchers couldn't tell if they were distributed evenly, clumped together, or breaking apart during manufacturing — all things that matter enormously for battery performance.

Making the invisible visible
The Oxford team developed a staining technique that attaches traceable silver and bromine markers to the cellulose and latex binders used in both graphite and silicon anodes. Once tagged, the binders show up clearly — they emit characteristic X-rays or reflect high-energy electrons that imaging equipment can detect.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News Detox"For the first time, we can accurately see the distribution of these binders not only generally, but also locally, as nanoscale binder layers and clusters, and correlate them with anode performance," said lead author Dr. Stanislaw Zankowski. The insight is straightforward but powerful: now researchers can watch what actually happens to binders during manufacturing, rather than guessing.
What they found was striking. Tiny differences in how binders are distributed can significantly affect how fast a battery charges and how long it survives. By tweaking the slurry mixing and drying steps during electrode production — essentially small manufacturing adjustments — the team lowered the internal ionic resistance of test electrodes by as much as 40%.
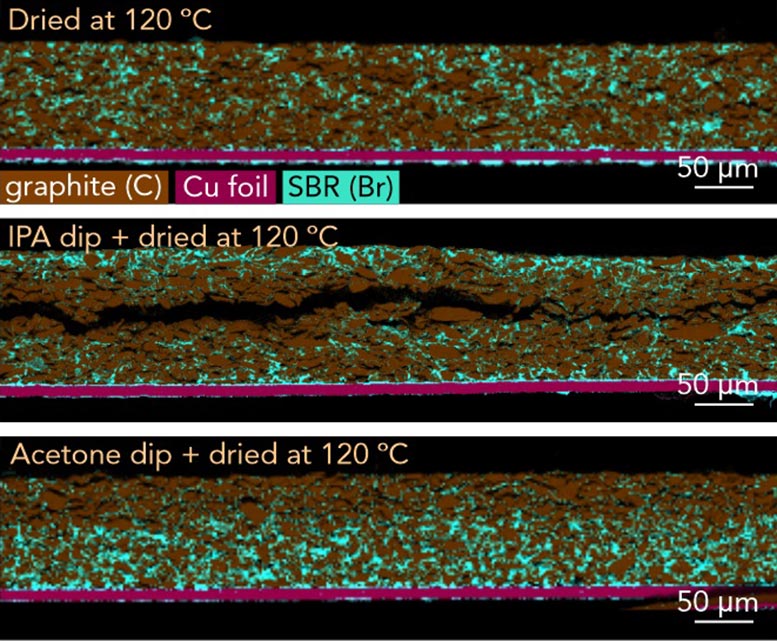
One particularly revealing discovery: a uniform coating of carboxymethyl cellulose binder on graphite particles can break apart into uneven, patchy fragments during processing. Those fragments weaken performance and long-term stability. Now that researchers can see this happening, they can prevent it.

The research, published in Nature Communications, has already caught the attention of major battery manufacturers and electric vehicle companies. The technique itself is patent-pending, and it's backed by the Faraday Institution's Nextrode project — a sign that this isn't just academic curiosity. The ability to see and optimize binders could translate into batteries that charge faster and last longer across everything from phones to electric cars. The next phase is watching whether these lab improvements scale up to real-world production.









