Imagine a world where the energy pouring from your solar panels or wind turbines doesn't just power your home, but gets neatly tucked away for a rainy, windless day. That's the promise of hydrogen fuel, a clean energy carrier that leaves no carbon footprint. The catch? Making it has been about as cheap and efficient as trying to catch smoke with a sieve. Until now.
A team led by Gang Wu at Washington University in St. Louis just cracked a major part of the code. They've found a new, surprisingly affordable way to split hydrogen from water, which could finally make large-scale renewable energy storage a reality. Because, let's be honest, those expensive platinum-group metals currently needed for the job were really putting a damper on the whole "affordable clean energy" thing.
The Catalytic Combo That Changes Everything
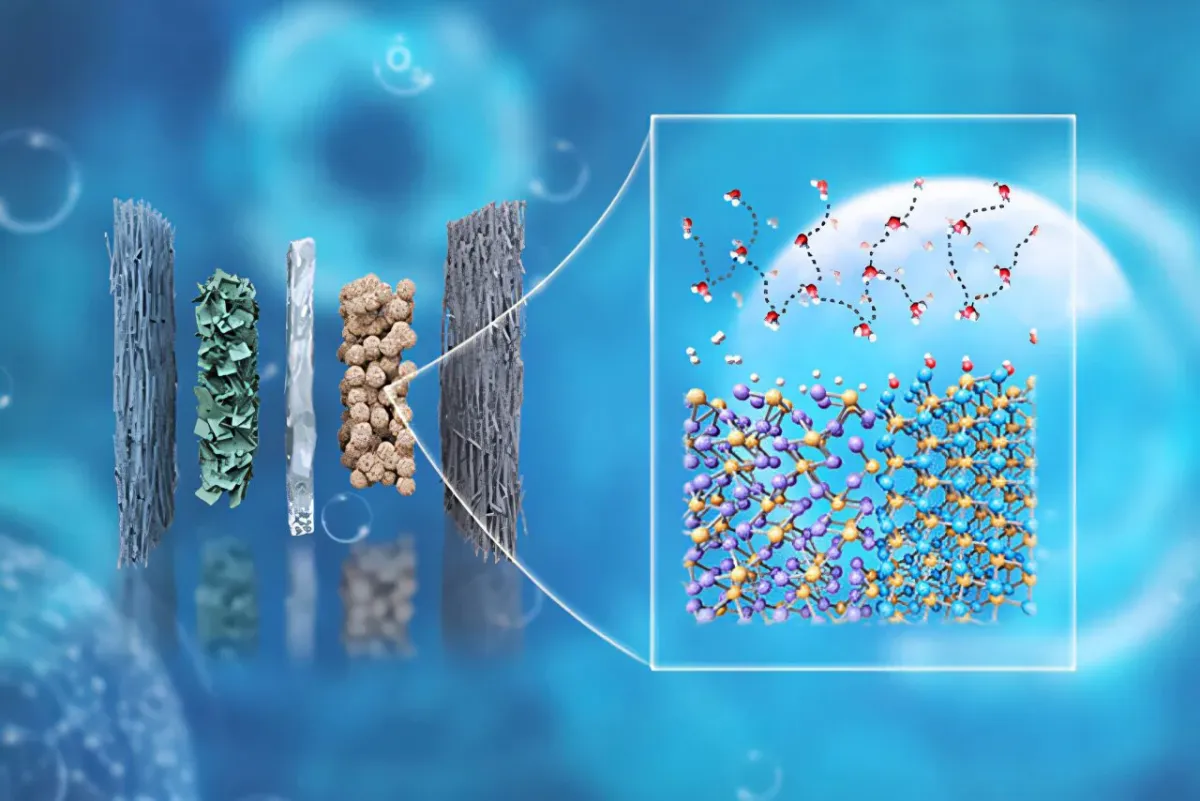
Their big breakthrough? A brand-new catalyst for anion-exchange membrane water electrolyzers (try saying that five times fast). This fancy system uses renewable electricity to, well, split water. But instead of relying on precious platinum, the new catalyst combines two phosphide materials: rhenium phosphide and molybdenum phosphide. Think of it as a dynamic duo where each material plays its part perfectly.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxRhenium helps hydrogen stick and unstick from the catalyst, while molybdenum speeds up the water-splitting process itself. When this clever combo was paired with a nickel-iron anode, the results were, frankly, impressive. The cathode didn't just perform better than other leading alternatives; it outshone the benchmark set by those pricey platinum-group metals.
And for the real kicker? It ran at industrial current densities for over a thousand hours. Let that satisfying number sink in. This makes it one of the most durable, platinum-free cathodes for this kind of electrolyzer out there. Which, if you think about it, is both impressive and slightly terrifying for anyone who just invested heavily in platinum mines.
Wu’s team isn’t just making better tech; they’re also figuring out why it works so well. They’re honing in on the crucial role of the hydrogen-bond network at the catalyst/electrolyte interface, which sounds complicated but essentially means they’re optimizing the molecular dance that makes hydrogen production so efficient. Their catalyst showed the lowest resistance, suggesting hydrogen is practically leaping off the surface.
While these experiments were conducted in a lab, the next step is to see if this game-changing cathode can scale up for industrial use. If it can, we might just be looking at a future where clean energy isn't just generated, but also stored, cheaply and efficiently, ready for whenever the sun takes a day off. And that's a future worth getting excited about.