Imagine building a tiny electronic component, not with wires and silicon, but with individual molecules. That's the microscopic dream scientists are chasing, and they just got a whole lot closer. Their latest creation? A five-in-one "super molecule" that could make our gadgets faster, smarter, and way more efficient.
The goal is to ditch the bulky stuff and build electronics one molecule at a time. The stars of this show are flat, carbon-rich molecules, which are surprisingly good at shuffling electrical charge around. Think of them as tiny, molecular-level expressways for electrons. We already use them in things like solar cells and chemical sensors, but the real prize is making them perform even better.
Now, the obvious move would be to link a bunch of these molecules together, creating a super-network that acts like one giant, powerful system. More connections, better electron flow, right? Faster everything. The problem? As these molecular chains get longer, they get stubborn. They stop dissolving in liquids, which makes them basically impossible to work with. It's like trying to build a LEGO castle underwater with pieces that keep floating away.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxThe Hybrid Hack
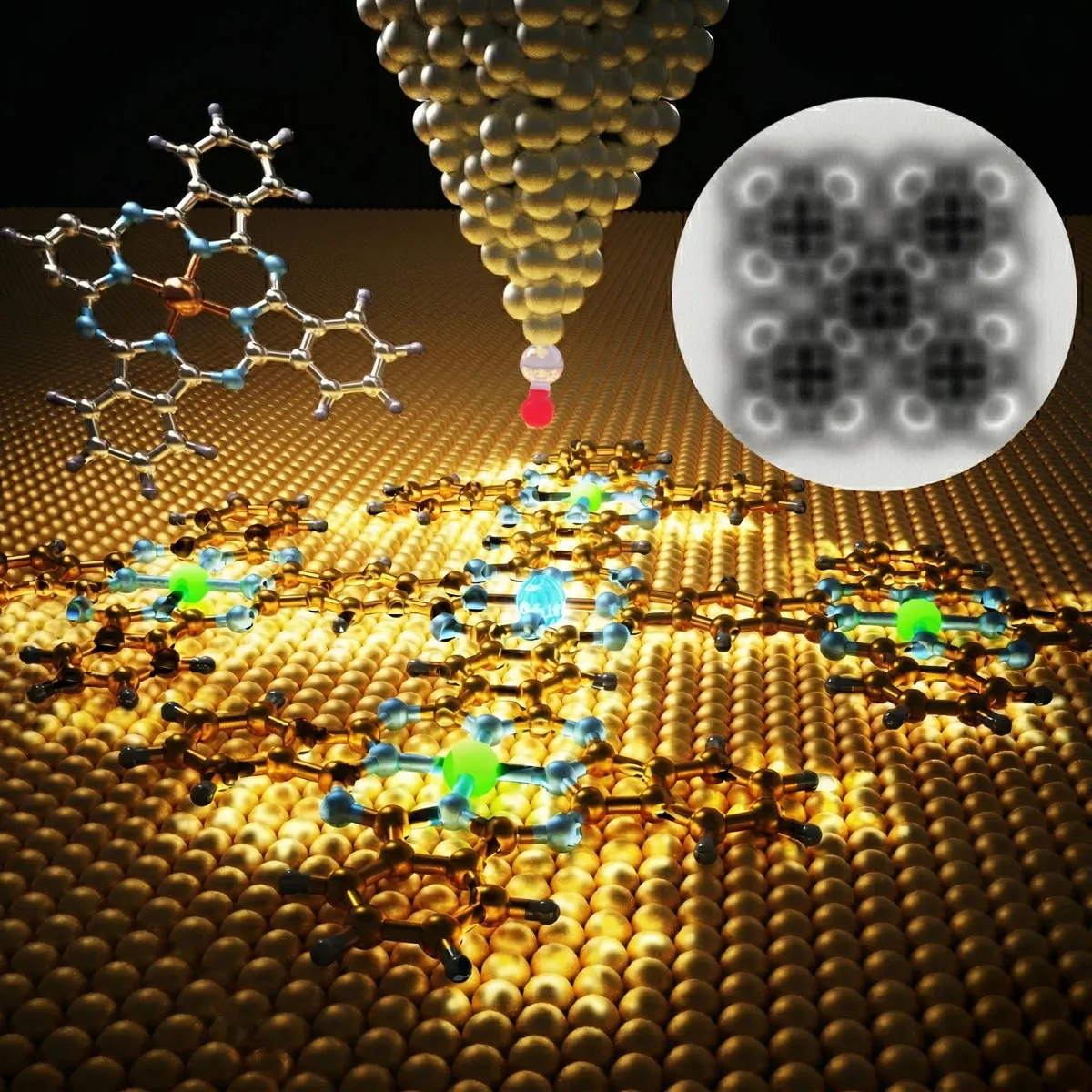
Enter the clever minds of Luis M. Mateo and Diego Peña at CiQUS. They found a workaround, a kind of chemical hack that combines two different approaches. First, they build special "phthalocyanine" units in a liquid, nice and easy. Then, they carefully place these pre-made units onto a metal surface. And here's where the magic happens: on that surface, the units react and snap together, forming a larger, cross-shaped structure made of five fused phthalocyanines. Suddenly, the impossible is, well, possible.
Mateo points out that the surface wasn't just a workbench; it helped create the structure and let them study it up close with special microscopes. Which, if you think about it, is both impressive and slightly terrifying. We're talking about manipulating matter at an atomic scale.
This new, cross-shaped molecule isn't just a pretty face. It acts as a single electronic system, and initial experiments show that linking the units together actually lowers the energy gap. In plain English? It makes it easier for charge to move, which is exactly what you want for high-performance materials.
Even cooler, these phthalocyanine units can hold tiny metal atoms in their centers. This means scientists can essentially drop different metals into specific parts of the structure, adding new properties, like magnetism, right in the middle of the molecule. Because apparently that's where we are now.
Peña's team is already dreaming bigger, looking to tweak the design to create actual two-dimensional polymers from these phthalocyanines. If they pull that off, we're talking about entirely new nanomaterials with properties we can barely imagine for molecular electronics, quantum tech, and energy systems.
So, the next time your phone charges faster or your solar panel generates more power, just remember: it might be thanks to a tiny, cross-shaped super molecule, quietly doing its job, one atom at a time.