Imagine a molecule so shy, so fleeting, that scientists have been theorizing about its existence for seven decades without ever actually seeing it. Well, that elusive wallflower, called tetroxide, has finally been caught on camera, so to speak. And its debut could change how we understand everything from air pollution to the very chemistry happening inside your body.
For 70 years, tetroxide was the Higgs boson of oxidation chemistry — a fundamental piece of the puzzle that everyone assumed was there, but no one could prove. Its existence was first floated in the 1950s as part of the Russell mechanism, a chemical tango where two unstable organic radicals briefly link up, forming a molecule with four oxygen atoms in a row. The catch? It vanishes in a fraction of a second.
The Unstable Star of the Show
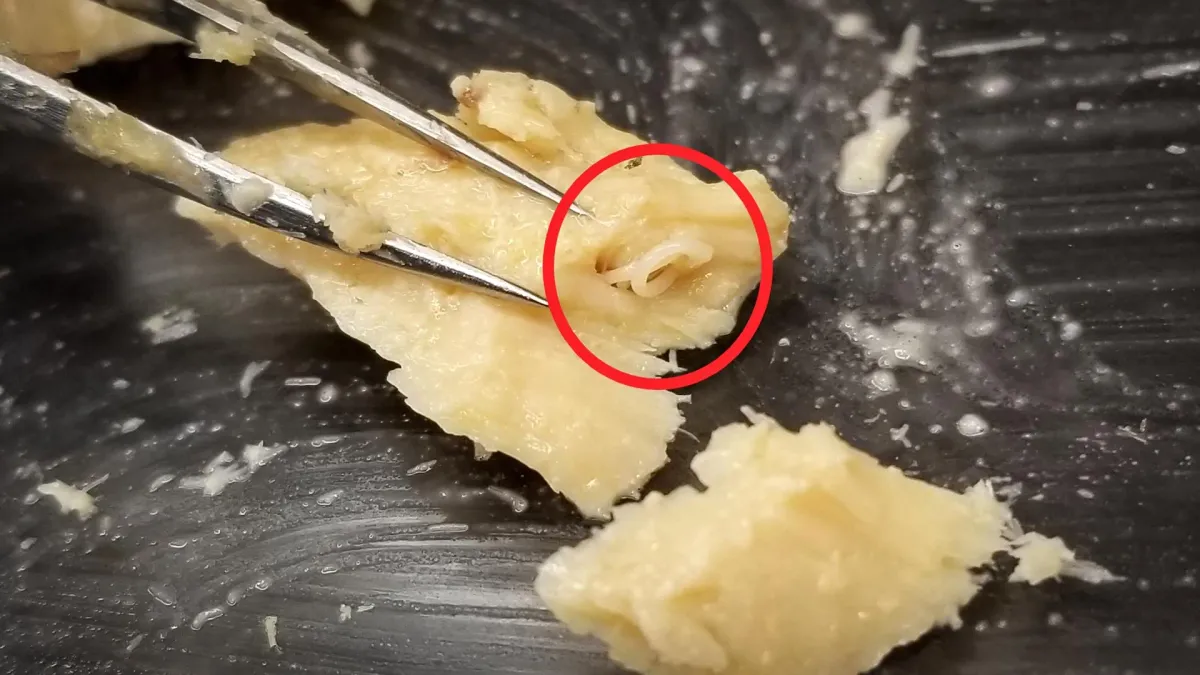
Researchers from KTH Royal Institute of Technology in Sweden and Kinetic Chemistry Research in California finally managed the impossible. They used advanced mass spectrometry, essentially a super-sensitive chemical weigh station, to snag these oxygen-rich molecules before they could poof into thin air. Because apparently, that's where we are now: inventing tech that can photograph ghosts.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxTetroxides are crucial to oxidation reactions, which are happening constantly, everywhere. They're involved in combustion, they break down pollutants in the atmosphere, and they're buzzing around inside you as we speak. But they also contribute to the less-than-glamorous side effects, like smog and those tiny airborne particles that make you cough.
Before this breakthrough, any evidence for tetroxides was either circumstantial or required freezing conditions so extreme, it'd make a polar bear shiver. The new method allowed scientists to observe them without, you know, accidentally breaking them apart in the process. Which, if you think about it, is both impressive and slightly terrifying.
Even more surprising? The team found that tetroxides are actually pretty stable in normal air, a stark contrast to what earlier, super-chilled experiments suggested. They can exist at room temperature, for a lifespan between 0.2 and 200 milliseconds. That's not exactly eternity, but it's long enough to get some serious chemical work done.
This discovery means tetroxides might be involved in all sorts of unknown reactions in outdoor environments and even living organisms. It could rewrite our understanding of how long pollutants (think paint solvents or smoke) linger in the air, and how other airborne compounds and aerosol particles form. It also opens new avenues for medical research, especially in areas like oxidative stress and potential cancer treatments. Because when you finally see a ghost, you realize it's been haunting more places than you thought.