Gardeners and farmers have long known that adding organic stuff to soil makes it hold water better. It's one of those things everyone just knows. But why? Turns out, scientists at Northwestern University finally cracked the molecular code, discovering a tiny, sticky secret that makes all the difference.
They found that carbohydrates — the same kind of stuff found in plants and microbes — act like a microscopic superglue. These carbs use water to form connections between organic materials and soil minerals, creating tiny bridges that trap moisture. We're talking about links so strong, they can keep soil hydrated even in bone-dry conditions. Which, if you think about it, is both impressive and slightly terrifying for anyone who's ever overwatered a houseplant.
This isn't just about your backyard, though. This discovery might also explain how water has managed to stay locked in rocks for billions of years, from ancient Earth to meteorites, and even on Mars. Because apparently, the Red Planet also needs a good, carbohydrate-rich compost.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxLudmilla Aristilde, who led the study, put it simply: the right mix of minerals and organic matter equals happy, moist soil. "Everyone has experienced it, but we haven't fully understood the physics and chemistry," she noted. Now that they do, she believes we could engineer soil to be like "long-term sponges" that refuse to give up their moisture.
The Sweet Science of Water-Trapping Bridges
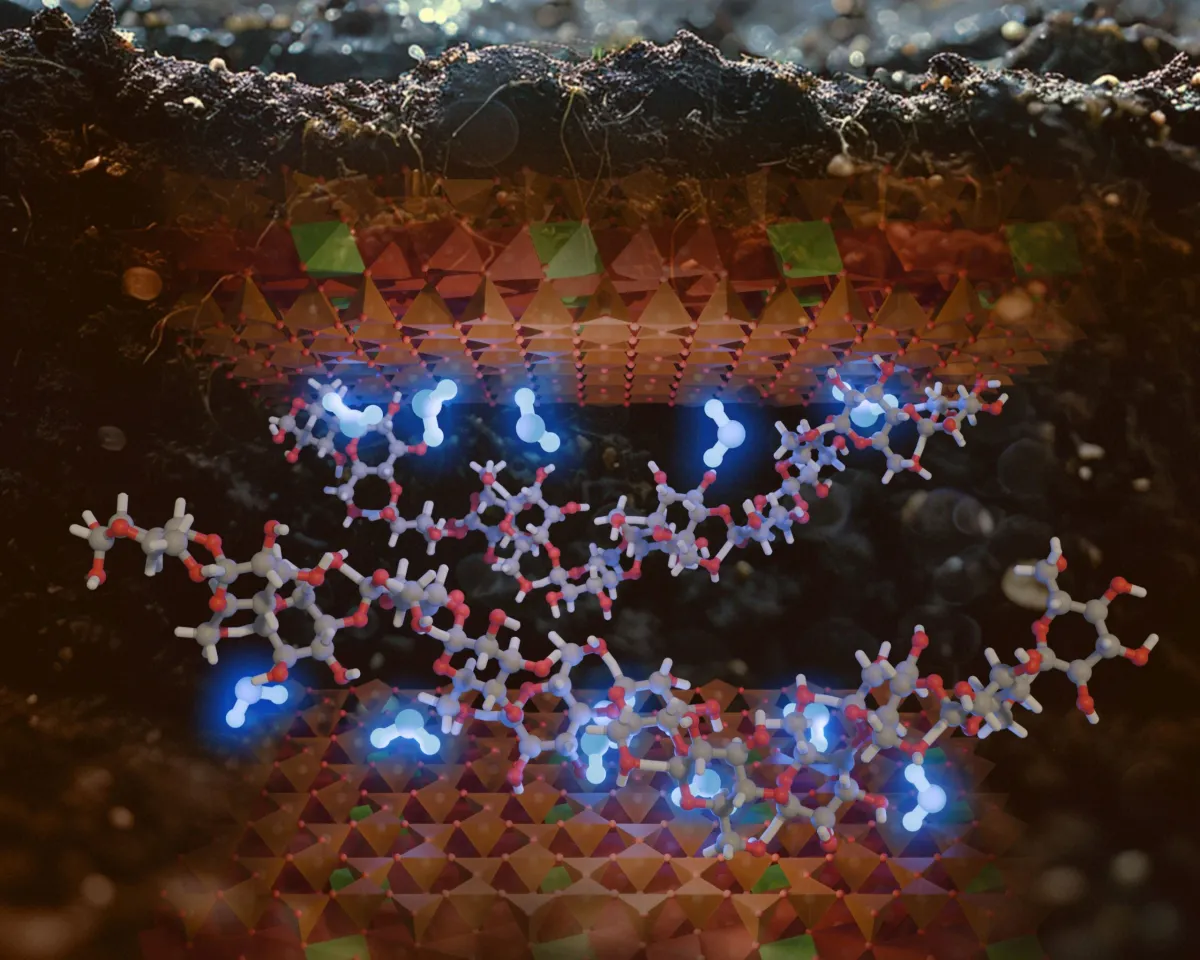
The team got down to the nitty-gritty, combining a common clay mineral called smectite with three types of carbohydrates: glucose (a simple sugar), and the more complex starches amylose and amylopectin. They picked these because carbs are everywhere, released by plants and microbes constantly, and their chemistry is just simple enough not to muddy the results.
Through computer simulations, calculations, and good old lab experiments, they zeroed in on hydrogen bonds. These are the weak forces that make water molecules cling to each other, like tiny, clingy magnets. The big discovery? Water molecules can bond to both clay surfaces and carbohydrates simultaneously. This creates little water bridges, connecting the two and holding moisture tighter than a toddler to a juice box.
Aristilde explained that when a water molecule is held by a hydrogen bond with a carbohydrate and a hydrogen bond with a mineral surface, its binding energy goes way up. It's essentially "stuck" between the two, making it much harder for that precious moisture to evaporate.
And it gets better: the simulations showed that water trapped this way had five times stronger binding energy than water attached only to clay. Even when things got seriously parched, this water was far less likely to escape. The team even cranked up the heat, finding that water required significantly higher temperatures to leave the mix when both clay and carbohydrates were present. Consider it the soil's ultimate hydration hack.
But wait, there's more. The structure of these complex carbohydrates also helps keep the clay's tiny pores open. Normally, these pores shrink and collapse as they dry out, kicking out any remaining moisture. But long, branched carbohydrate molecules act like microscopic scaffolding, preventing the pores from collapsing completely. This means soils can hold onto moisture for much longer, even when a drought decides to move in for an extended stay.
So, next time you're marveling at a lush garden or wondering how Mars might have once supported life, remember the humble carbohydrate. It's the unsung hero, the molecular glue, making the world (and maybe other worlds) a little bit wetter, one tiny bridge at a time.