Cancer cells in the same tumor don't all behave the same way. Some grow fast, others slow. Some resist drugs, others don't. For years, scientists studying how cancer cells communicate have hit a wall: their tools only showed them the average picture, mixing signals from millions of cells at once. That blur has just gotten sharper.
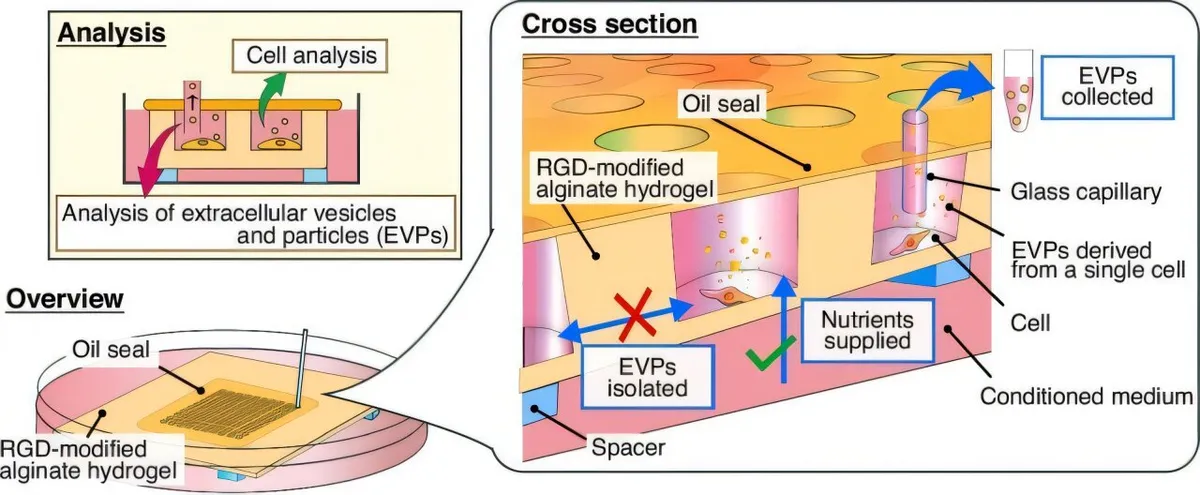
Researchers at Keio University have built a microdevice that does something previously impossible — it lets individual cancer cells live for nearly three weeks in sealed microwells while capturing every message they send out. Those messages come in the form of extracellular vesicles, tiny packets of proteins and RNA that cells release to talk to their neighbors. By trapping these vesicles right at their source, the team can finally see which cell said what.
The engineering is elegant. Each microwell holds a single cell in a space so small that nutrients can flow in to keep the cell alive, but the vesicles it releases can't escape. It's like a cell-sized greenhouse with a one-way filter. Over 19 days, the researchers watched individual cancer cells grow at wildly different rates, even though they all came from the same cell line. More importantly, they collected the vesicles each cell released and analyzed them separately.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxWhat they found matters. Some cells pumped out dozens of vesicles; others released just a handful. The size and protein composition of those vesicles varied dramatically from cell to cell. "This level of resolution is critical for understanding why cells that look similar can behave so differently," the researchers noted. You can't see that variation when you're averaging across a million cells.
This isn't just a technical win for the lab. The ability to link vesicles back to their exact source cell opens a clearer view into how cancer spreads, why some tumors resist treatment, and which cells are actually the troublemakers. It also works for spotting biomarkers — the molecular signatures doctors use to diagnose disease and predict outcomes. The platform could eventually track RNA and proteins from the same single cells over time, building a more complete picture of how individual cells change.
Scaled up, this approach could analyze hundreds or thousands of cells in parallel, moving medicine away from population averages toward something more precise: understanding the actual behavior of the cells that matter most.