For nearly 20 years, orbital data from Mars showed something that shouldn't be there: layers of iron sulfate with a spectral signature no one could explain. Now a team led by Dr. Janice Bishop at NASA's Ames Research Center has solved the mystery, and the answer rewrites when Mars stayed geologically alive.

The key is a mineral that forms only under heat. In laboratory experiments, scientists heated common Martian sulfates to above 100°C and watched them transform into ferric hydroxysulfate—a compound with a crystal structure so distinct it may be entirely new to science. The catch: Mars's surface today is frozen. So if this mineral exists there now, something had to have been heating it in the recent past.
-Map-of-Valles-Marineris-Region-777x429.jpg)

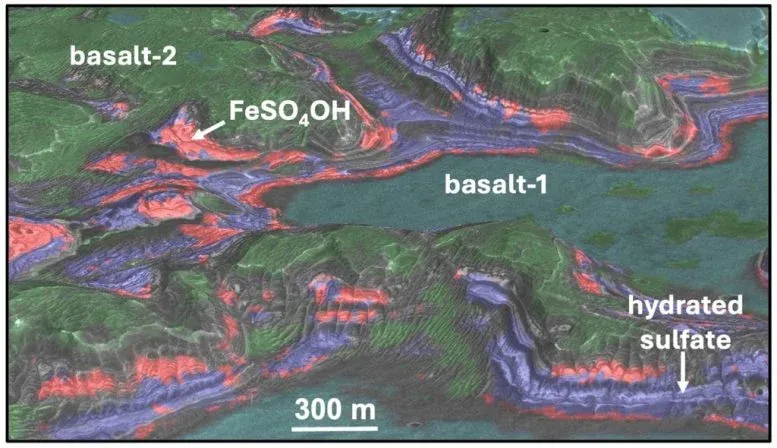
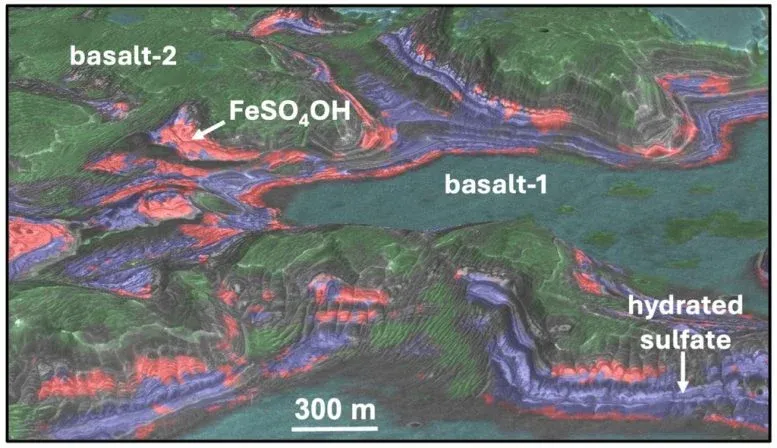
The deposits sit in two regions near Valles Marineris, Mars's vast canyon system. One is Aram Chaos, where ancient water once flowed. The other is a plateau above Juventae Chasma, a canyon five kilometers deep. Both preserve the scars of Mars's wetter past. But the sulfate layers here tell a different story: they formed after the planet's warm, wet period ended, buried beneath basaltic rock in a way that suggests volcanic heat or geothermal energy worked on them later.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News Detox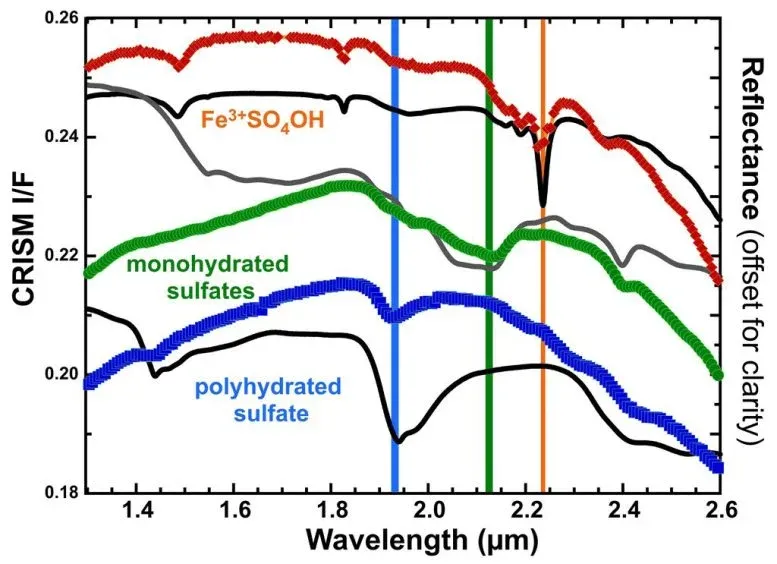
What makes this significant is the timeline. The sulfates likely transformed during the Amazonian period, which ended less than three billion years ago. That's recent in geological terms—recent enough that it suggests Mars wasn't the dead, frozen world we've long imagined. Volcanic activity at the Juventae Plateau and geothermal energy beneath Aram Chaos could have provided the heat needed for these mineral conversions, meaning parts of Mars were still chemically and thermally active long after its golden age of surface water.

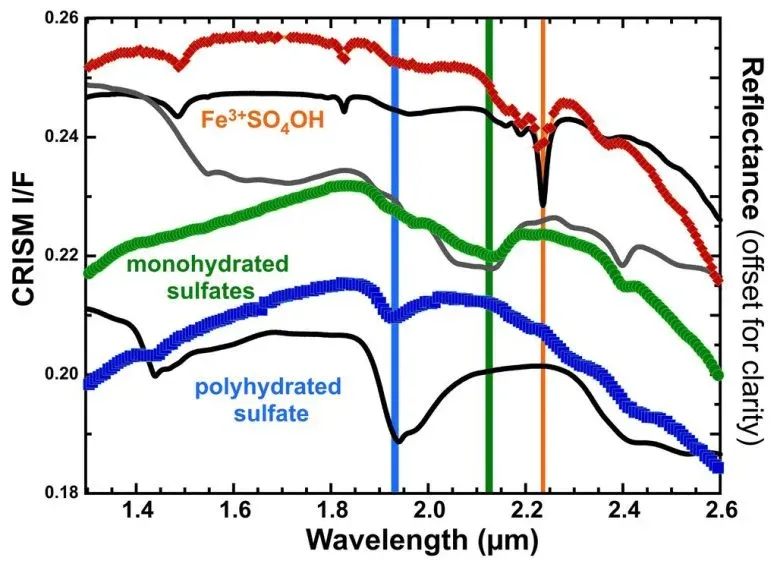
Dr. Johannes Meusburger, a postdoctoral researcher on the team, explained the chemical chain reaction in the lab: rozenite (a sulfate with four water molecules) heats to become szomolnokite (one water molecule), then further heating produces ferric hydroxysulfate, where hydroxyl groups replace the remaining water. The whole process requires oxygen and releases water vapor. Mars's thin carbon dioxide atmosphere still contains enough oxygen for this to work.
The discovery opens a window into Mars's later history that we've largely overlooked. We've known the planet had water and warmth early on. We've known it cooled and dried. But the evidence for persistent geothermal activity—for heat persisting in the subsurface long after the surface froze—changes how we think about Mars's potential to harbor life. Subsurface heat can drive chemistry and sustain microbial ecosystems even when the surface is hostile. If Mars had geothermal energy available three billion years ago, the conditions for life underground may have persisted far longer than surface evidence alone would suggest.

Bishop noted that while the material formed in the lab is likely a new mineral—it has a unique crystal structure and thermal stability—scientists must also find it on Earth to officially recognize it as a new mineral. That's a quirk of mineralogy: even if you synthesize something in the lab, it doesn't count as a true mineral until nature has made it somewhere. The discovery, published in Nature Communications, now gives other researchers a target to search for, both on Mars and potentially in Earth's geothermal zones.
The next phase is understanding how widespread this geothermal activity was. If it occurred in multiple regions, it suggests Mars's interior remained active longer and more extensively than current models predict. That would reshape not just our understanding of Mars's geology, but our thinking about where life might have persisted on a cooling world.










