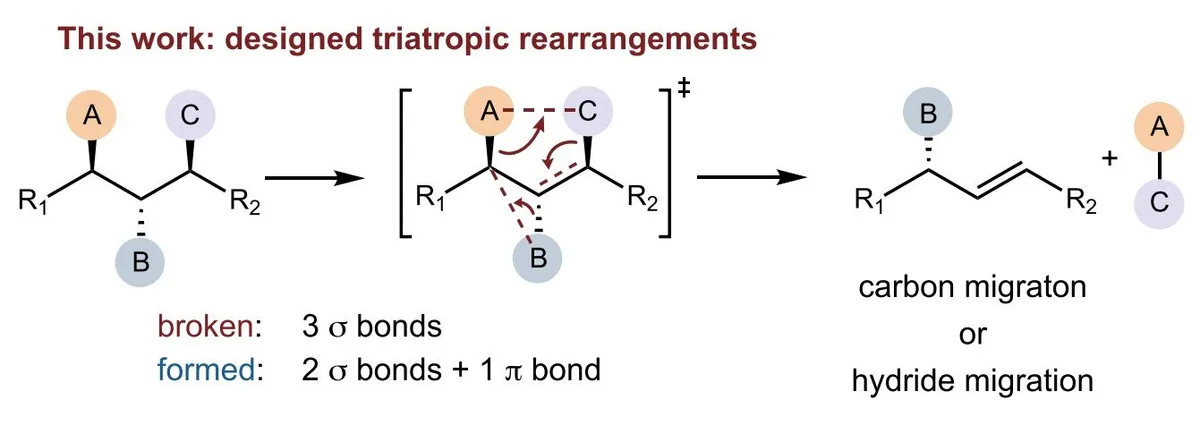
Synthetic chemists have a new tool for one of their most frustrating problems: reshaping molecules by changing the size of their carbon rings. Researchers at Southern University of Science and Technology have discovered a reaction that does something previously difficult in a single, precise step—breaking three bonds and rebuilding them as two bonds plus one double bond, all at once.
The breakthrough, published in Science, centers on what's called a triatropic rearrangement. The mechanism lets chemists shrink six-membered rings down to five-membered rings, a transformation that matters because even tiny changes in ring size can completely alter how a molecule behaves—its strength, its reactivity, how it interacts with other substances.
Triatropic rearrangements provide synthetic tools for late-stage reshuffling of saturated rings through boron-enabled transmetallation with zinc salts. Credit: Science (2026). DOI: 10.1126/science.adw3340
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxWhy this matters
Older methods like the Favorskii and Wolff rearrangements could achieve similar ring transformations, but they were messy—hard to control, and they often produced unwanted side products. This new approach combines the efficiency of pericyclic reactions (reactions that happen in one smooth, coordinated motion) with a simple elimination step. Using organoboron and zinc reagents as helpers, chemists can now convert epoxides—three-membered rings—into cyclopentanes—five-membered rings—with remarkable precision and selectivity.
The reaction gives chemists three things they need: control over which bonds get broken and where new ones form (chemoselectivity), control over which part of the molecule reacts (regioselectivity), and control over the 3D orientation of the final product (stereoselectivity). It also leaves behind a useful chemical handle—an alkenyl boronic ester—that can be modified further, opening doors to building more complex structures.
For drug discovery and materials science, this matters because faster, more reliable ways to reshape molecules means faster iteration. Instead of spending weeks troubleshooting a finicky ring-shrinking reaction, chemists can move forward with better tools. The precision also reduces waste and failed attempts, which saves time and resources.
The work signals how incremental advances in reaction chemistry compound. One cleaner tool in the synthetic toolkit becomes the foundation for the next discovery. This particular mechanism is already being explored for applications in pharmaceutical synthesis and advanced material design.