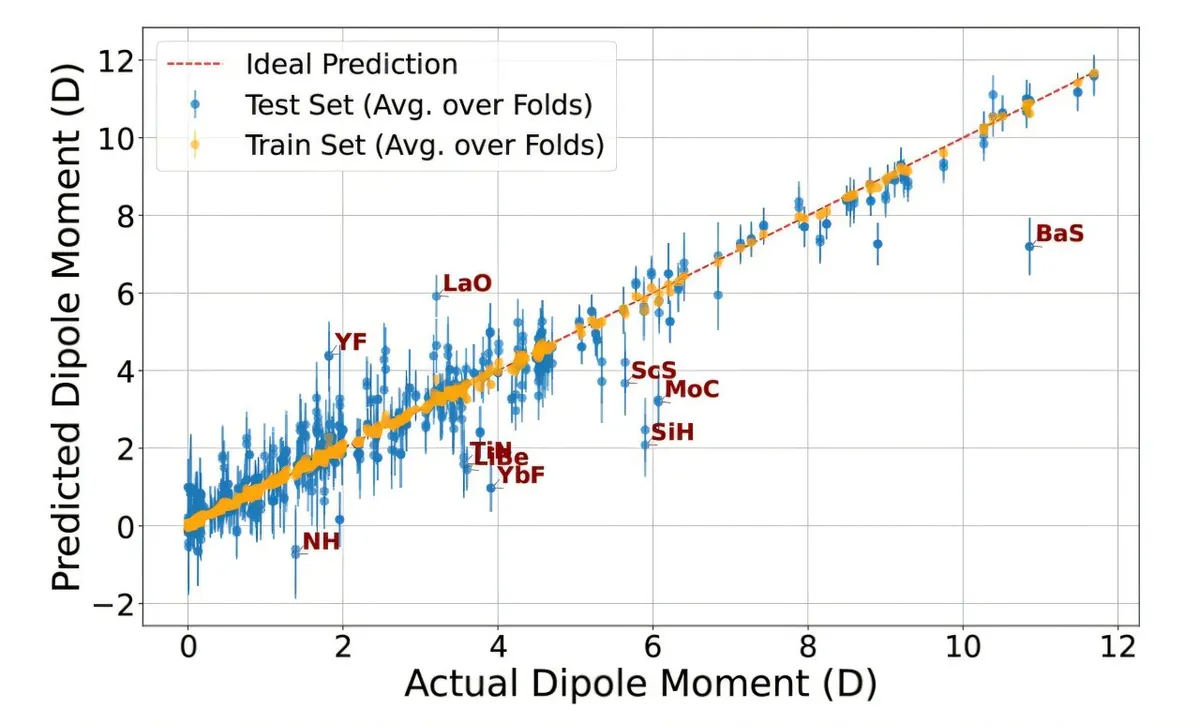
Chemists just got a seriously cool new shortcut. Scientists built an AI that can predict how strongly an electric charge is split in a molecule – in mere seconds. All it needs are the basic properties of the atoms involved.
Think of it like this: every molecule has a positive and a negative end. How far apart those charges are is called its "dipole moment." It’s basically a molecule’s unique fingerprint. This tiny detail changes everything from how hot something needs to get to boil to how well it conducts heat.
Understanding these dipole moments is a big deal for everything from basic chemistry to developing new tech. And this AI just flipped the script on how we find them.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxThe AI's Wild Discoveries
This new AI uses something called Gaussian Process Regression. It zipped through over 4,800 molecules, nailing their dipole moments with crazy accuracy. It didn't just confirm what we knew; it found some wild new candidates.
We're talking super heavy, salt-like molecules like cesium iodide (CsI) and francium iodide (FrI). But here’s the kicker: it also found surprising pairs, like gold–cesium (AuCs), that traditional chemistry wouldn't have predicted to have such strong dipoles.
Usually, figuring out a molecule's dipole moment is a slow process, needing expensive lab gear or complex computer models. Older AI models needed a bunch of info about the molecule itself – info that’s often missing for new discoveries. But this new model is different.
It only uses simple, direct facts about the atoms: how much they like electrons, how much energy it takes to pull an electron off, and where they sit on the periodic table. It learned from a dataset of 273 molecules, then went to work on thousands more.
It pinpointed molecules with dipole moments above 11 Debye – that’s like finding the strongest magnets in a pile of regular ones. The biggest ones were in heavy alkali halides (think CsI) and even alkali–gold molecules. This AI can even tell you which molecule will have the strongest dipole for any given atom.
Beyond Old Rules
For ages, chemists thought a molecule's dipole was mostly about how "greedy" an atom was for electrons (its electronegativity). But the AI showed that's not the whole story. Relying only on electronegativity can actually lead you astray.
The model proved that other factors, like how easily an atom loses or gains electrons, are super important for getting it right. For instance, the AI found that metals like gold (Au) can act like non-metals when paired with certain atoms, creating unexpectedly strong dipoles.
Scientists are buzzing about these strong-dipole molecules, especially for "cold molecular sciences." They act in unique ways in quantum environments, which is pretty nuts. This model could seriously speed up the hunt for new molecules, maybe even helping us explore physics beyond what we currently understand. Seriously cool stuff.