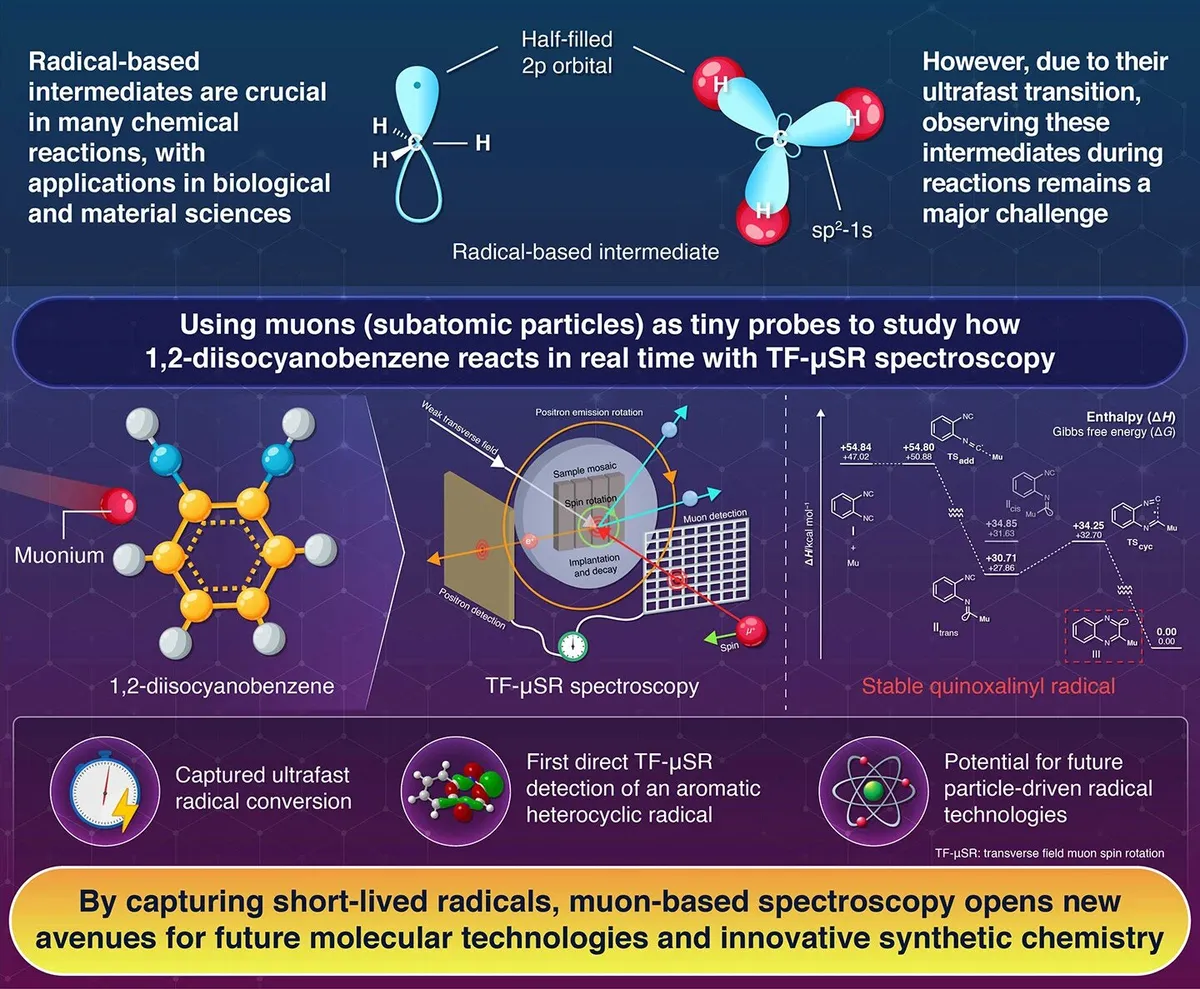
Chemists have long faced a frustrating problem: the molecules they need to study disappear in billionths of a second. Now, researchers from Japan and Canada have developed a way to actually watch these fleeting chemical intermediates transform in real time.
The breakthrough uses an unconventional tool—muons, subatomic particles that decay into positrons. When a muon captures an electron, it forms muonium, a light version of hydrogen. By tagging molecules with this muonium marker, scientists can track what happens as reactions unfold at the atomic scale.
Associate Professor Shigekazu Ito and his team at Tokyo Tech, working with researchers at TRIUMF in Canada, used a technique called transverse field muon spin rotation spectroscopy (TF-μSR) to observe something that had never been directly seen before: an imidoyl radical instantly collapsing into a quinoxalinyl radical. The entire transformation took just a few nanoseconds—billionths of a second.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News Detox"We wanted to visualize the ultra-fast radical processes that are fundamental in organic chemistry," Ito explains. "TF-μSR allowed us to follow the imidoyl radical as it converted into a quinoxalinyl radical, uncovering a cyclization that happened in mere nanoseconds."
Why does this matter? Radicals—highly reactive chemical fragments—are essential to everything from drug design to new materials. But because they exist for such impossibly brief moments, chemists have mostly had to infer their behavior rather than observe it directly. This new technique opens a window into that hidden world.
The quinoxalinyl radical the team observed turned out to be remarkably reactive, quickly stealing hydrogen atoms from surrounding solvent molecules. Understanding exactly how and why these reactions happen could help chemists design better drugs, more efficient catalysts, and novel electronic materials.
The research sets a new benchmark for what's possible in radical chemistry. As the technique becomes more refined, it could let chemists predict and control these ultra-fast reactions with unprecedented precision—accelerating innovation in synthetic chemistry and molecular engineering.