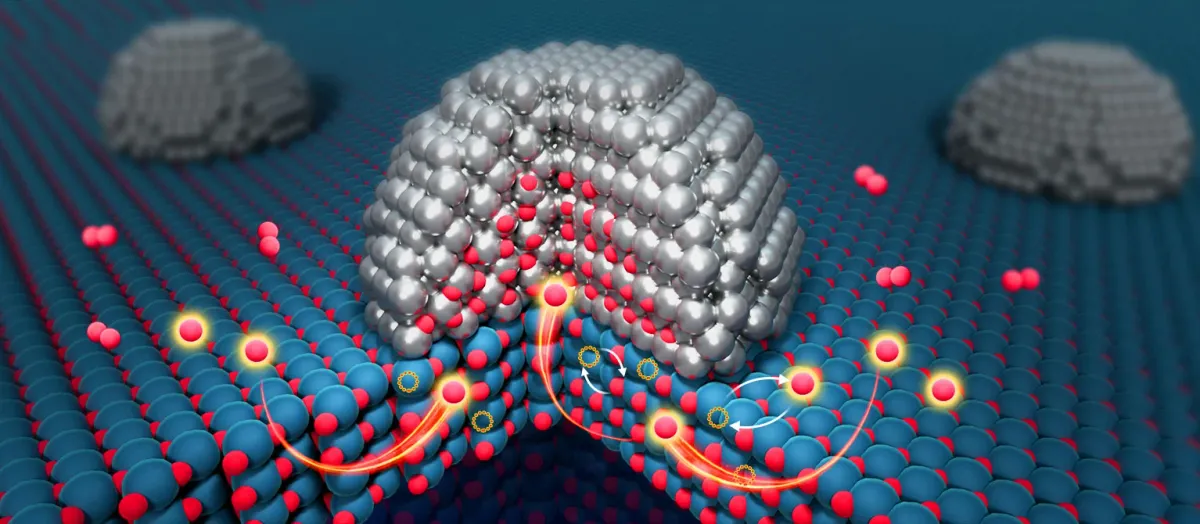
A new study has uncovered a hidden pathway inside catalysts. This pathway allows oxygen to move through deeper parts of the material, not just its surface. This discovery could change how chemical reactions are designed.
Scientists led by Tao Zhang and Yanqiang Huang at the Dalian Institute of Chemical Physics, along with Wei Liu and Yanggang Wang, tracked oxygen movement in catalysts. Their findings challenge long-held beliefs about how these materials work.
Oxygen Moves Through Catalyst Interiors
Using advanced microscopy, the team saw oxygen moving through the inside of a catalyst in real-time. This is the first direct evidence of such movement. The results, published in Nature, suggest that parts of catalysts once thought inactive might actually be important.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxCatalysts are vital for many industries, speeding up reactions for things like fuel and pollution control. A key process is "spillover," where atoms like oxygen move between a metal and its support material.
For decades, scientists thought this movement only happened on the outer surface. This idea influenced how catalysts were designed, focusing on maximizing surface area.
Now, researchers show that oxygen atoms can travel through the main body of the material. This opens up a new area for chemical activity.
Visualizing the Movement
To see this, the team used titanium dioxide (TiO2), a material known for storing and releasing oxygen. It's used in things like solar energy and environmental cleanup.
They combined TiO2 with ruthenium, a metal often used in catalysts. This allowed them to watch oxygen atoms move at an atomic level.
Instead of staying on the surface, oxygen atoms moved from layers three to five atoms deep within the TiO2 towards the metal. This movement is driven by differences in oxygen chemical potential, which pulls oxygen through the material.
The study also found that the boundary between the metal and the support material controls this movement.
Wei Liu explained that TiO2 has a channel for oxygen spillover. The metal-support interface acts like a "guard," controlling if oxygen can pass. This suggests a new way to use the bulk of a catalyst, which was previously thought to be useless.
New Ways to Design Catalysts
Professor Yanqiang Huang noted that this oxygen spillover allows the bulk of a catalyst to help with mass transfer during reactions. This highlights how important it is to engineer interfaces to control spillover.
These findings build on decades of research into how metals and support materials interact. About 50 years ago, scientists found that metal particles could be covered by oxide supports like TiO2. This reduced their ability to absorb molecules. Traditionally, these interactions were thought to happen only on the outer surfaces where the metal and support meet.
This new study expands that idea. It shows that oxygen spillover can also happen through internal interfaces within the catalyst. These internal areas, normally out of reach for reacting molecules, can still help move material during reactions.
The results show how important it is to carefully design catalyst interfaces. They also demonstrate how advanced imaging can reveal reaction pathways at the particle level.
The team plans to keep developing this approach. Tao Zhang said they can improve catalysis from two-dimensional surface reactions to a three-dimensional "surface–interface–bulk" system. This offers new insights into how to engineer interfaces in catalysts. The next goal is to create practical catalysts that use the bulk material directly in chemical reactions.
Deep Dive & References
Imaging interface-controlled bulk oxygen spillover - Nature, 2026