Your fingertip knows the difference between a light tap and a hard press. Your arm knows when you're being stretched. And until recently, science had no idea why.
It turns out the answer lives in the molecular architecture of your cells—specifically, how a protein called PIEZO2 is physically wired to the scaffolding inside them.
Researchers at Scripps Research have been studying PIEZO2 for years. It's one of two nearly identical proteins that act like tiny doors in your cell membranes, opening when mechanical force arrives and letting charged particles flood in. Those particles create electrical signals your brain reads as touch, pain, or body position. The puzzle was this: PIEZO2's close relative, PIEZO1, responds to broad stretching forces (like when cells in your blood vessels expand). But PIEZO2 is exquisitely sensitive to small, localized pressure—the kind of delicate sensation you need to feel a handshake or a raindrop.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxWhy the difference, when the two proteins look almost identical under a microscope?
The Wiring That Changes Everything
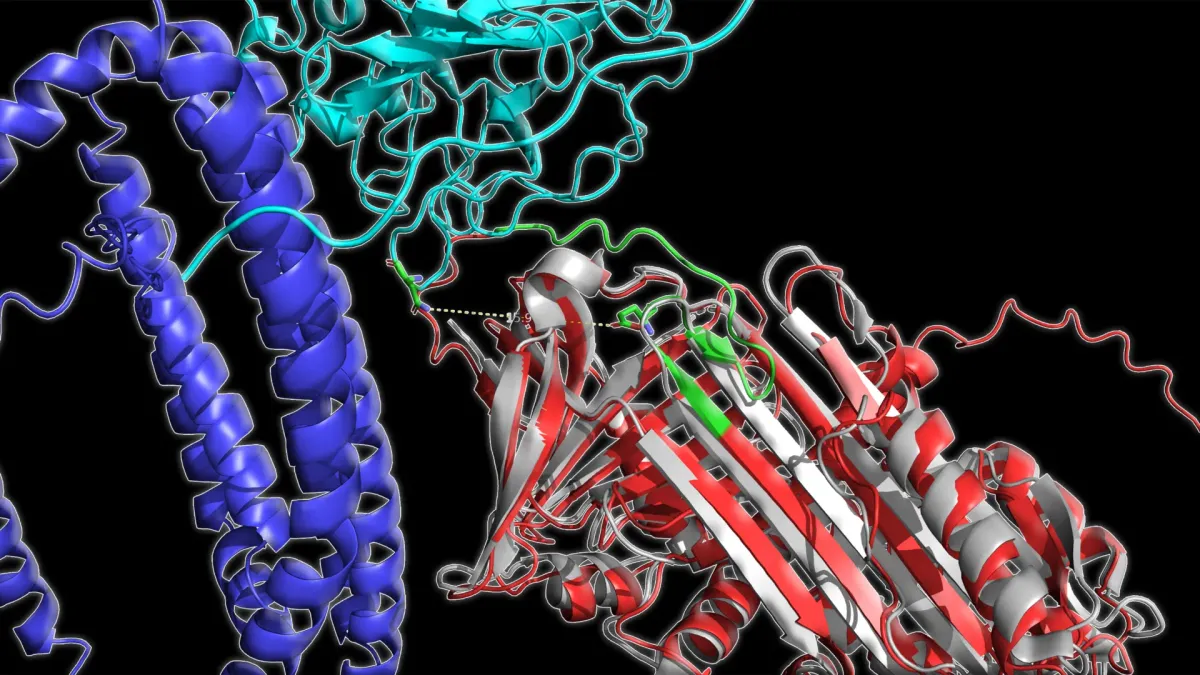
The team used an imaging technique called MINFLUX super-resolution microscopy—think of it as a 100,000x more precise camera than standard methods—to watch PIEZO2 actually moving inside living cells. What they found was striking: PIEZO2 isn't floating freely in your cell membrane. It's tethered to the cell's internal skeleton, a network of protein fibers called the actin cytoskeleton, through a connector protein named filamin-B.
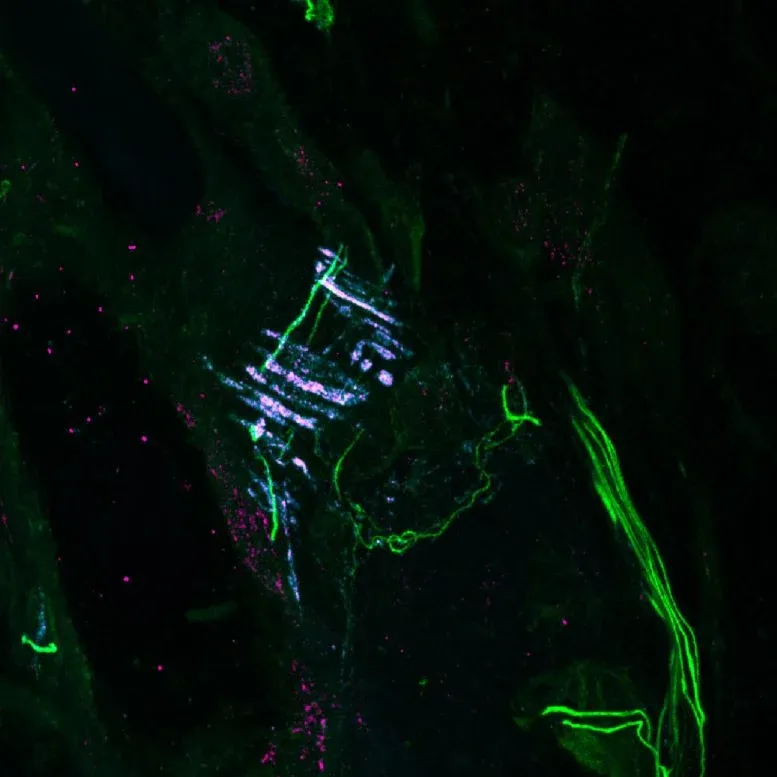
That tether is the whole story. When your skin gets poked, the force travels through the cell's scaffolding directly to PIEZO2, making it far more likely to open. But when a cell simply stretches—the kind of force that activates PIEZO1—that same tether actually prevents PIEZO2 from responding. It's like the difference between pushing a doorbell directly versus pushing on the wall next to it.
When the researchers removed the tether in mouse sensory neurons, PIEZO2 suddenly behaved like PIEZO1, responding to stretch instead of pressure. "We were surprised by how differently the two channels responded to the same type of force," recalls Eric Mulhall, the study's first author. The implication is wild: your cells fine-tune what they can feel not just by choosing which protein to use, but by controlling how that protein is physically connected to everything around it.
Why This Matters Beyond Your Fingertips
Mutations in PIEZO2 cause sensory disorders that affect how people feel touch and sense their body's position in space. Mutations in filamin-B are linked to skeletal and developmental conditions. By mapping out exactly how these proteins work together, the research provides a clearer framework for understanding what goes wrong when they don't.
"A protein's physical connections inside a cell determine what kinds of forces it can sense," says Ardem Patapoutian, the study's senior author and a Nobel Prize winner for his earlier PIEZO discoveries. "That's a new way of thinking about how we feel the world around us."
The findings suggest that touch isn't just about having the right protein—it's about how that protein is anchored, oriented, and wired into the larger machinery of the cell. Your skin's ability to feel a whisper instead of a shout comes down to engineering at the scale of billionths of a meter.