Imagine being able to turn a protein on or off just by raising the temperature in a specific cell by a few degrees. That's no longer theoretical. Researchers at Heidelberg University have developed a way to engineer proteins that respond to subtle heat changes, opening a path toward far more precise control of cellular processes without invasive intervention.
The breakthrough centers on something called allosteric thermoswitches — essentially molecular on-off buttons that flip at specific temperatures. The team, led by Prof. Dr. Dominik Niopek and Dr. Jan Mathony, integrated heat-sensitive domains from plants into various proteins, allowing them to respond with precision to temperature shifts within the narrow window where human cells naturally operate (37°C to 40°C). The work was published in Nature Chemical Biology.
Why this matters
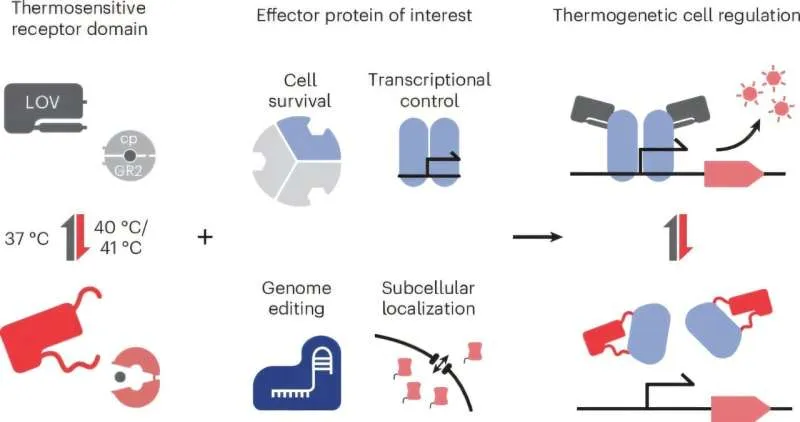
Until now, controlling proteins with temperature was considered impractical — most methods worked indirectly, by switching genes on and off rather than controlling the proteins themselves. That limitation meant scientists couldn't fine-tune cellular behavior in real time. Heat, though, has real advantages as a control signal. It penetrates tissue deeply, can be applied non-invasively, and doesn't require chemicals that might interfere with other cellular processes.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxThe Heidelberg team solved the engineering problem by taking a plant sensory protein and optimizing it to work inside mammalian cells. They tested the concept first in bacteria, then moved to human cell cultures, where they built temperature-controlled versions of CRISPR gene editors. The results showed they could activate or deactivate these tools with precision by adjusting temperature by just a few degrees.
"Using these allosteric thermoswitches, we are able to directly and reversibly control cellular functions without actively intervening in other processes of the cell," explains Prof. Niopek. This distinction matters: many current tools for controlling cells require adding chemicals or turning genes on and off, both of which can have unintended side effects.
The modular breakthrough
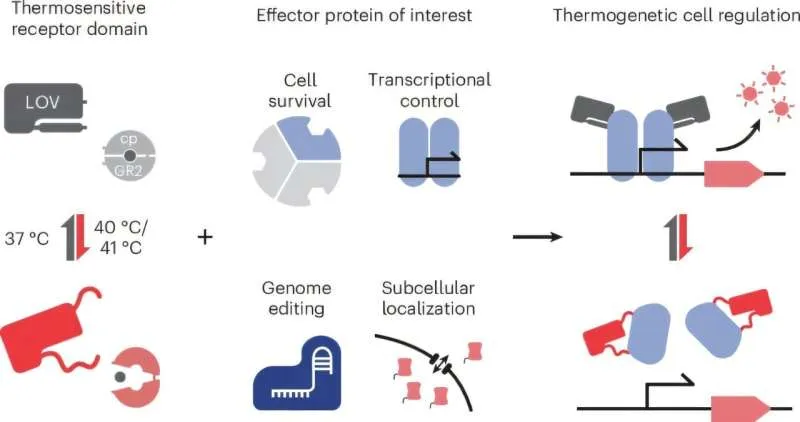
What makes this work genuinely novel is its modularity. The researchers didn't just create one heat-controlled protein — they developed a design blueprint that can be applied to almost any protein, regardless of what it does or how it's shaped. They've even shown the approach works with multiple different heat-sensing domains, meaning the system is flexible and adaptable.
This is the kind of foundational tool that tends to have ripple effects. Right now, it's being tested on CRISPR editors. But the same principle could be applied to control immune cells, regulate enzyme activity, manage gene expression, or even coordinate complex cellular responses in tissues. Anywhere a researcher needs precise, reversible control without chemical side effects, this could eventually work.
Dr. Jan Mathony notes that the field is "at the threshold of the possible" — meaning the core concept is proven, but scaling it to practical applications is the next phase. The team is already working on making thermogenetics more comprehensive and broadly applicable, with an eye toward future biomedical uses.