Imagine a molecule so unstable it essentially evaporates the moment it touches water. For decades, scientists thought trying to stabilize one in H2O was, well, bonkers. Yet, a team of chemists just did exactly that, confirming a vitamin B1 theory from 1958 and potentially paving the way for greener medicine.
The Case of the Vanishing Carbene
At the heart of this molecular magic trick is a 'carbene.' Think of it as a carbon atom that's missing two electrons, making it incredibly reactive. Most carbon atoms prefer a cozy eight electrons, but carbenes only have six. This means they're not just unstable; they're aggressively unstable, reacting with pretty much anything in their immediate vicinity. Especially water, where they typically disintegrate upon contact.
Back in 1958, a Columbia University chemist named Ronald Breslow proposed that vitamin B1 (thiamine) might briefly morph into a carbene-like structure inside cells. This fleeting transformation, he theorized, would be crucial for key biochemical reactions. The problem? No one could ever actually see it. It was too unstable, too ephemeral, particularly in the watery environment of a living cell.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxThe 'Suit of Armor' Solution
Fast forward 67 years, and a team led by UC Riverside chemistry professor Vincent Lavallo has not only created a carbene that stays stable in water but isolated it. They bottled the thing, and it remained intact for months. Lavallo, clearly enjoying the vindication, noted that people once thought this idea was "crazy," but Breslow was, in fact, absolutely right.
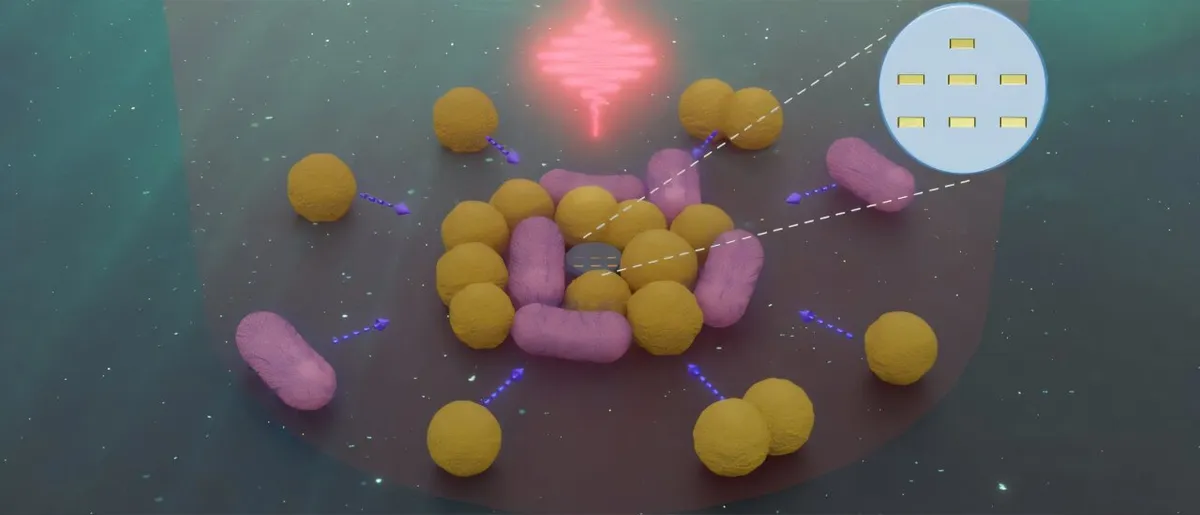
Their secret? A molecular "suit of armor." Lavallo's team built a protective structure around the carbene, shielding its reactive core from water and other molecules nearby. This allowed them to study it with advanced techniques like nuclear magnetic resonance spectroscopy and x-ray crystallography, providing irrefutable proof that these molecules can, indeed, exist in water.
Greener Meds and Cellular Mimicry
This isn't just about proving an old theory, though that's satisfying enough. Carbenes are often used as 'ligands' — supporting actors in metal-based catalysts that drive crucial chemical reactions, from making medicines to fuels. The catch? These processes typically rely on toxic organic solvents.
By stabilizing carbenes in water, researchers have unlocked a path toward significantly safer and more eco-friendly chemical production. As Varun Raviprolu, the first author on the study, put it, "Water is an ideal solvent: abundant, non-toxic, and environmentally benign." Getting these powerful catalysts to work in water is a giant leap for greener chemistry.
Beyond that, being able to create and maintain reactive molecules in water brings scientists closer to mimicking the complex chemistry that happens naturally inside living cells, which are, after all, mostly water. Lavallo believes this protective method could finally allow scientists to isolate and study other reactive intermediates that have, until now, remained unseen.
For Lavallo, who's worked with carbenes for two decades, this is a profound milestone. Thirty years ago, people doubted these molecules could even be made. Now, they're bottled in water, confirming a 'crazy' idea from the 1950s. It just goes to show: never underestimate a good theory, even if it takes a few decades (and a molecular suit of armor) to prove it.