Imagine your cells as tiny, bustling factories. Inside, there’s a crucial organelle called the endoplasmic reticulum (ER) — basically, the protein production line and calcium storage unit. Now, what happens when this vital bit gets damaged? It doesn’t just sit there. Your cells have a remarkably elegant, if slightly gruesome, solution: ER-phagy. It's a fancy term for “ER self-eating,” where damaged parts are gobbled up by double-membraned sacs called autophagosomes.
For a long time, scientists were scratching their heads, wondering how this cellular cleanup operation actually kicked off. What was the signal? Where did those hungry autophagosome membranes even come from?
The Cell’s Own Motion Sensors
Enter a research team led by Zhang Hong from the Chinese Academy of Sciences. They just cracked the code, and it turns out the whole thing starts with some specialized ion channels — essentially, tiny motion sensors inside your cells. These channels detect mechanical changes, then use calcium signals to initiate the breakdown of damaged ER. Because apparently, even your cellular components have a highly sophisticated “check engine” light.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
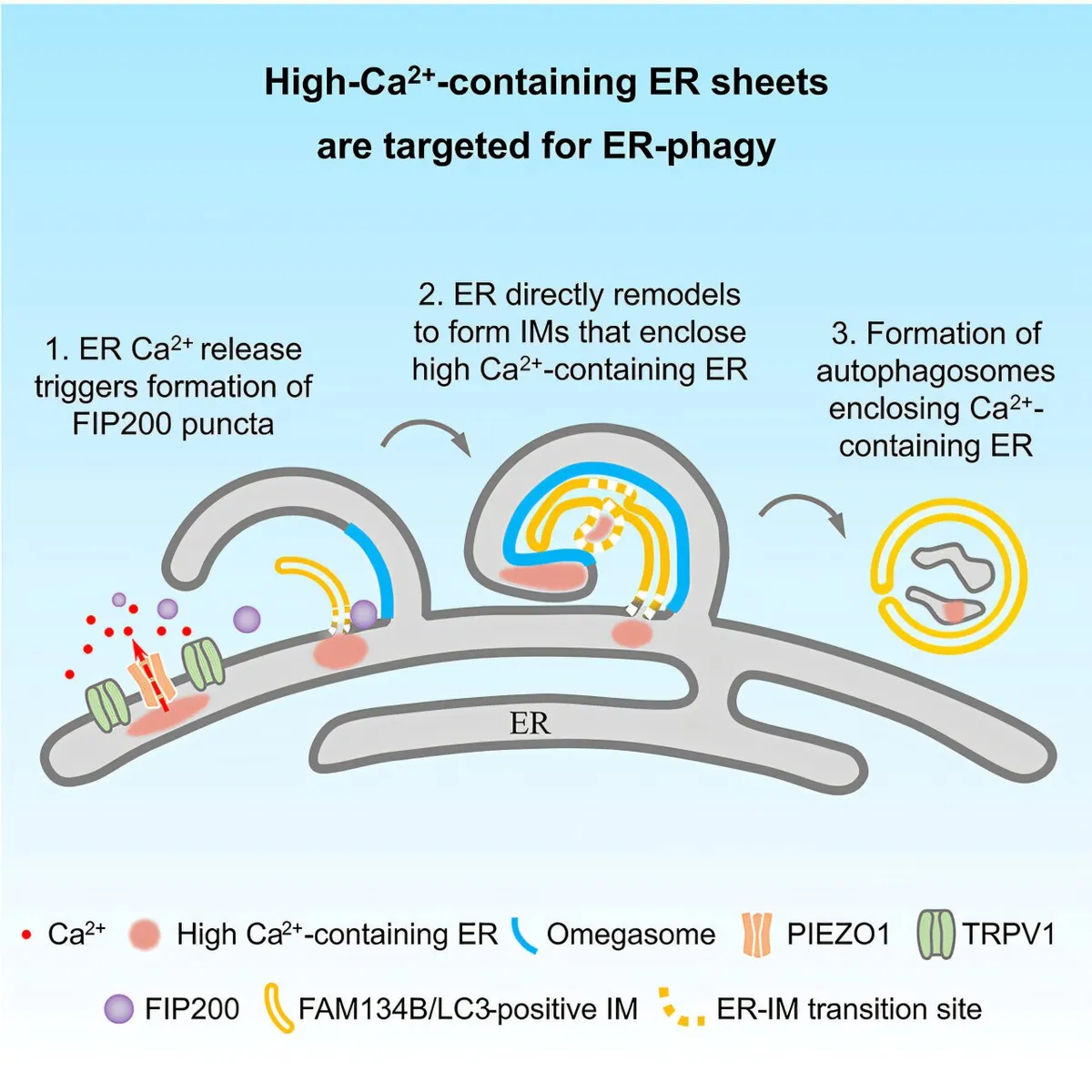
Start Your News DetoxPublished in Molecular Cell, their study used advanced imaging to literally watch cells under stress. What they saw was fascinating: parts of the ER, specifically those rich in calcium, would swell up. Then, like tiny Pac-Men, autophagosomes would move in, surround, and dismantle these calcium-laden sections. Electron microscopy even showed that the membranes for these cleanup crews came directly from the ER itself. Talk about recycling!
The team pinpointed two key players in this cellular drama: the ER-phagy receptor FAM134B and lipidated LC3. But the real stars of the show were two mechanosensitive channel proteins, PIEZO1 and TRPV1. These little guys hang out in those calcium-rich ER areas, detect changes in calcium levels, and then release calcium locally, creating quick, targeted signals.
These calcium bursts then cause a complex called FIP200 (the autophagy initiator) to separate into liquid-like droplets, which, in turn, triggers the whole ER-phagy process. It’s like a microscopic Rube Goldberg machine, but for cellular health.
Why This Matters (Beyond Cellular Housekeeping)
Disruptions in ER calcium balance and ER-phagy are linked to a whole host of nasty conditions, from brain disorders to various cancers. By finally figuring out how stress-damaged, calcium-rich ER sections are flagged for removal, this study provides a much clearer roadmap to understanding — and potentially treating — these diseases. Suddenly, those tiny cellular motion sensors don’t seem so small after all.