Every second of every day, tiny electrical charges zoom through your body. They're the silent heroes of life, making sure your cells chat, churn out energy, and keep your metabolism purring. And it all hinges on the meticulous movement of positive charges, called protons.
Enter phosphoric acid. It's the unsung MVP found everywhere from your DNA to the energy currency in your cells (ATP). It's also a star player in batteries and fuel cells because, well, it's really, really good at moving those protons around.
But how does it do it? Protons don't just free-range through these molecules. Instead, they play a game of hopscotch, leaping from one molecule to the next, guided by molecular pathways called hydrogen bonds. Scientists call this "proton-shuttling," and it's how charges zip around at lightning speed.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxWe've known about this shuttling for ages, but the nitty-gritty molecular dance remained a mystery. So, researchers from the Fritz Haber Institute teamed up with partners from Leipzig and the United States to get a closer look at the very first steps of this proton transfer.
The Coolest Molecule on Earth (Almost)
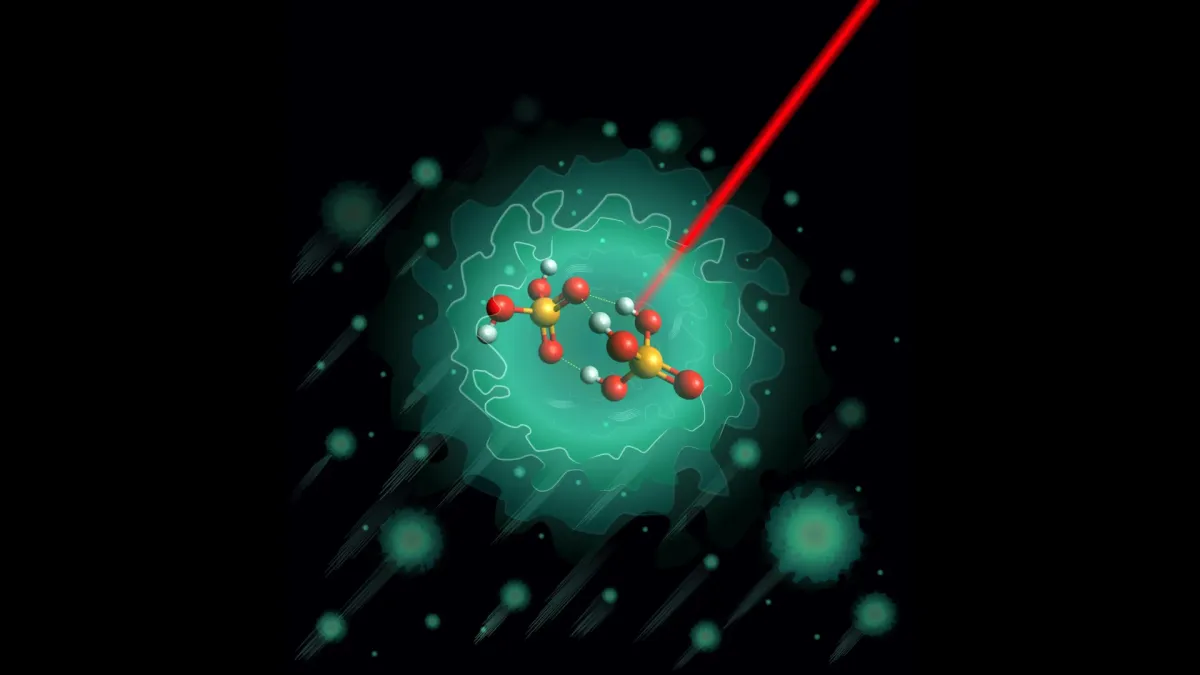
Previous research hinted that a specific negatively charged molecule, the deprotonated dimer H3PO4·H2PO4-, was the key. So, the scientists cooked up this molecule in the lab and then decided to give it the ultimate chill-out session.
They tucked it into a helium nanodroplet and cranked the cold down to a mind-boggling 0.37 degrees above absolute zero. That's colder than space, folks. At these extreme temperatures, almost all molecular jitters vanish, allowing them to precisely map its structure using infrared spectroscopy. Think of it as putting the molecule in a deep freeze to take its portrait.
They then cross-referenced these frosty findings with quantum chemical calculations, which predict how molecules arrange themselves. The combined approach painted a much clearer picture.
What they found was a bit of a plot twist. Theoretical models had predicted two equally likely structures for this molecule. But the experiments? They showed only one stable form. Just one. This lone structure turned out to be surprisingly rigid, featuring three hydrogen bonds locked together around a shared oxygen atom. It also had high barriers that made it tricky for protons to move within it. This suggests that this tight, interconnected setup might be a common feature in phosphoric acid clusters.
It's a stark reminder that even the most advanced theoretical models can miss crucial details, making good old-fashioned experimental testing absolutely essential.
Why This Matters to Your Future Fuel Cell
This discovery helps explain why phosphoric acid is such a spectacular proton conductor—earning it the nickname "Nature's proton highway." By pinning down a single, definitive structure for that key anionic dimer, scientists now have a much better handle on how protons navigate these systems.
This isn't just a win for molecular nerds. These findings provide a golden reference point that can help refine quantum chemical models and, crucially, guide the creation of new materials with even better proton conductivity. That's big news for the future of fuel cells and other energy technologies. Plus, it deepens our understanding of how proton transfer keeps all living things, including you, ticking along.
Now, go tell your DNA. It'll appreciate it.