How a tiny molecule became nature's charge carrier
Every electrical signal in your body—every heartbeat, thought, and muscle contraction—depends on charged particles moving through cells at precisely the right moment. Phosphoric acid, a molecule so small you'd need a microscope to see it, is one of the main conductors of this traffic. It's everywhere: the backbone of your DNA, the energy currency of your cells, the reason fuel cells can power cars. And for decades, scientists have known it was exceptional at moving electrical charge. They just didn't understand exactly how.
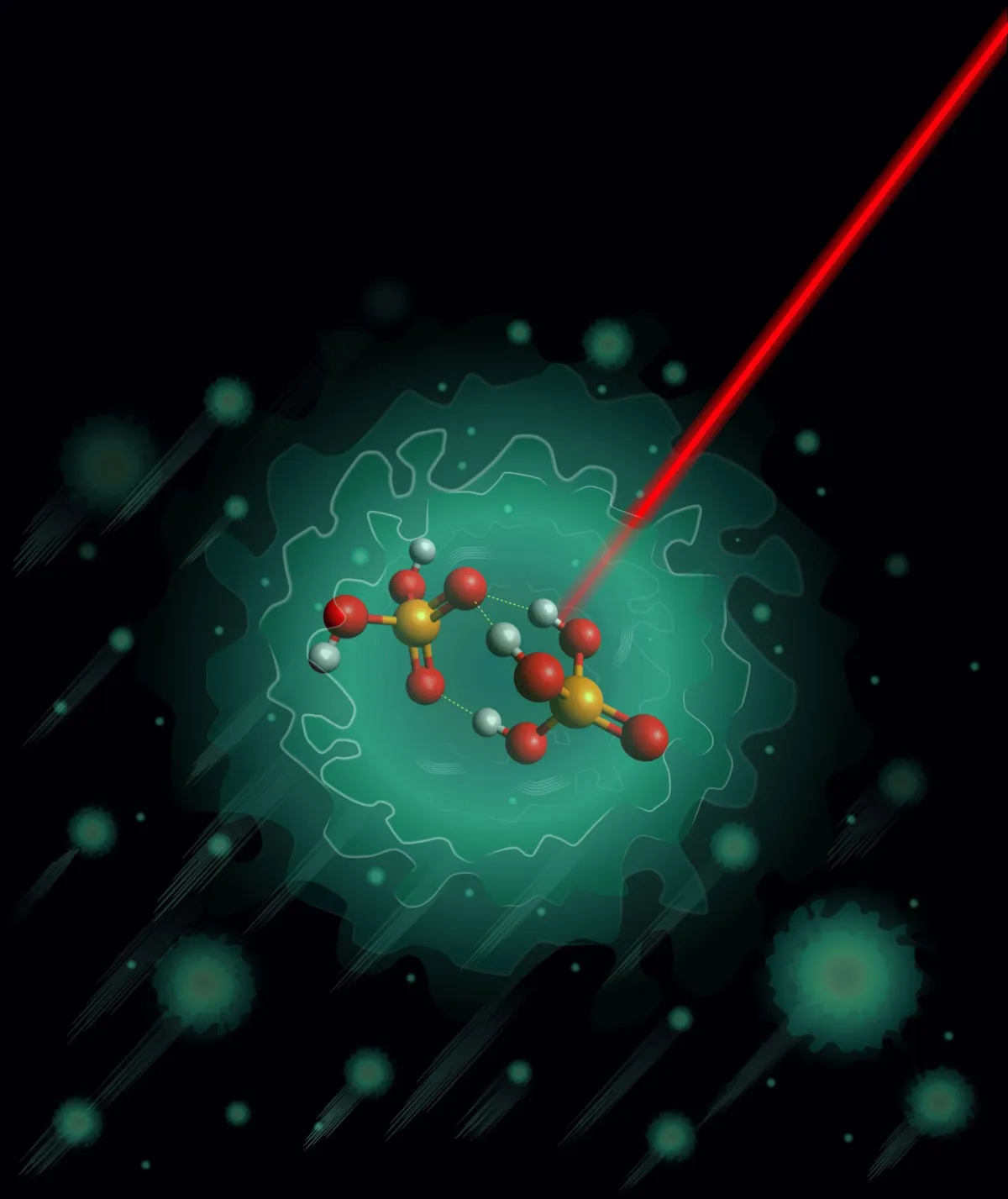
Researchers at the Fritz Haber Institute in Berlin have now filled in a crucial gap. By cooling a phosphoric acid complex to 0.37 degrees above absolute zero—colder than outer space—they watched it settle into a single, stable structure. That structure is the starting point for what happens next: protons jumping along a molecular relay, one bond to the next, in a process so efficient it works both in living cells and in the batteries we're building to power the future.
The experimental detour
For years, theoretical models predicted that this particular phosphoric acid structure should exist in two equally likely forms. But theory and reality don't always align. The team embedded their molecule inside a helium nanodroplet and cooled it down—extreme conditions that freeze out thermal noise and let you see the molecule's true shape. When they measured it with infrared radiation and compared the results to their predictions, something unexpected emerged: there was only one stable form, not two.
We're a new kind of news feed.
Regular news is designed to drain you. We're a non-profit built to restore you. Every story we publish is scored for impact, progress, and hope.
Start Your News DetoxIt's a small discovery with a large implication. The structure they found contains three hydrogen bonds arranged in a particular pattern, with a shared oxygen atom at the center. This same pattern shows up in other phosphoric acid clusters, suggesting it's a fundamental feature of how these molecules organize themselves. The finding also reveals something humbling: computational models, no matter how sophisticated, sometimes need the real world to correct them.
Why this matters for what comes next
Proton transport isn't just a biological curiosity. Engineers are already using phosphoric acid's proton-conducting properties in fuel cells—devices that could replace fossil fuel engines. Better understanding of the molecular structures involved means better materials, more efficient devices, and deeper insight into how life itself manages the constant flow of electrical signals that keeps everything running.
The work doesn't solve the puzzle of proton transport entirely. But it places a crucial landmark in the map. The next researchers who build on this foundation will know exactly where to look.